Advertisements
Advertisements
प्रश्न
Based on the group valency of element write the molecular formula of the following compound giving justification:
Oxide of first group elements.
उत्तर
Oxides of the first group elements have the common formula of M20.
Example- Na20, K20. This is because, the first group elements have a common valency of 1, and the valency of Oxygen is 2 so, to satisfy the combining capacity of Oxygen two 1st group metals are required. 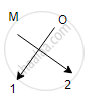
APPEARS IN
संबंधित प्रश्न
Two elements ‘A’ and ‘B’ belong to the 3rd period of Modern periodic table and are in group 2 and 13, respectively. Compare their following characteristics in tabular form:-
(a) Number of electrons in their atoms
(b) Size of their atoms
(c) Their tendencies to lose electrons
(d) The formula of their oxides
(e) Their metallic character
(f) The formula of their chlorides
Define the term : Atomic size
How do atomic structures (electron arrangements) change in a period with increase in atomic numbers from left to right?
Why does the size of the atoms progressively become smaller when we move from sodium (Na) to chlorine (Cl) in the third period of the periodic table?
The atomic numbers of the elements Na, Mg, K and Ca are 11, 12, 19 and 20 respectively. The element having the largest atomic radius is:
(a) Mg
(b) Na
(c) K
(d) Ca
Select the correct answer
Number of shells in potassium are
An element Z has atomic number 16. Answer the following
(a) State the period and group to which Z belongs
(b) Is Z a metal or a non-metal?
(c) State the formula between Z and hydrogen.
(d) what kind of a compund is this?
The elements of one short period of the periodic table are given below in order from left to right:
| Li | Be | B | C | O | F | Ne |
To which period do these elements belong?
What is the common feature of the electronic configuration of the elements at the end of Period 2 and Period 3?
Complete the table pertaining to the following elements given in Column I.
| Column I Elements | Atomic Number | Electronic Configuration | Underline the element present in that group |
| 1. Hydrogen | 1 | ______ | Be/Na/Ca |
| 2. Carbon | 6 | ______, ______ | N/P/Si |
| 3. Nitrogen | 7 | _______, ______ | C/P/S |
| 4. Oxygen | 8 | _______, ______ | N/S/Cl |
| 5. Sulphur | 16 | _____, ______, _______ | O/N/F |
| 6. Chlorine | 17 | _____, ______, ______ | I/O/S |
