Advertisements
Advertisements
प्रश्न
When phenolphthalein is added to NaOH, the colour of the solution will become _________.
- colourless
- red
- pink
- yellow
उत्तर
pink
When phenolphthalein is added to NaOH, the colour of the solution will become pink
APPEARS IN
संबंधित प्रश्न
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
Complete and balance the following chemical equations
Na2 CO3 (s) + HCI (aq) →
Which element is common to all acids?
Name the gas evolved when zinc granules are treated/heated with:
hydrochloric acid solution
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Name X and Y
What would be the colour of litmus in a solution of sodium carbonate?
State the common and chemical names of the compound formed when plaster of Paris is mixed with water.
Name the product formed when Cl2 and H2 produced during the electrolysis of brine are made to combine.
What is the chemical formula of bleaching powder?
State one use of bleaching powder (other than bleaching).
When _______________ is passed through fresh lime water, it turns milky.
Lime water reacts with chlorine to form:
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
Which of the following salts does not contain any water of crystallisation?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
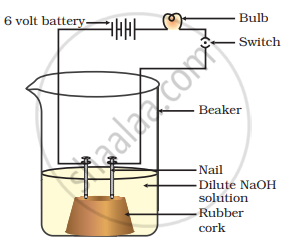
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids.
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
A sulphate salt of Group 2 element of the Periodic Table is a white, soft substance which can be moulded into different shapes by making its dough. When this compound is left in open for some time, it becomes a solid mass and cannot be used for moulding purposes. Identify the sulphate salt. Why does it show such a behaviour? Give the reaction involved.
A salt may be ______.
The formula of bleaching powder is ______.
On placing a copper coin in a test tube containing green ferrous sulphate solution, it will be observed that the ferrous sulphate solution ______.
