Advertisements
Advertisements
प्रश्न
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
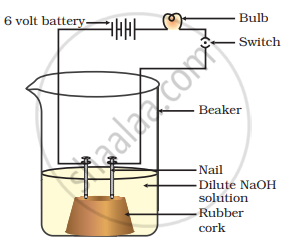
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
पर्याय
(i) and (iii)
(ii) and (iv)
(ii) only
(iv) only
उत्तर
(ii) only
Explanation -
Bulb will glow because the acid will furnish ions which conduct electricity.
APPEARS IN
संबंधित प्रश्न
Complete and balance the followingchemicalequations:
NaHCO3 (S) + HCI (aq) →
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
What are esters? How are esters prepared? Write the chemical equation for the reaction involved. What happens when an ester reacts with sodium hydroxide? Write the chemical equation for the reaction and also state the name and use of this reaction.
Calcium phosphate is present in tooth enamel. Its nature is
Common salt besides being used in kitchen can also be used as the raw material for making
- washing soda
- bleaching powder
- baking soda
- slaked lime
A sulphate salt of Group 2 element of the Periodic Table is a white, soft substance which can be moulded into different shapes by making its dough. When this compound is left in open for some time, it becomes a solid mass and cannot be used for moulding purposes. Identify the sulphate salt. Why does it show such a behaviour? Give the reaction involved.
What is the correct procedure for mixing acid and water? Why?
The metal that will not produce hydrogen gas when reacted with dilute acids.
Give a balanced equation for the reaction:
Silver nitrate solution and sodium chloride solution.
