Advertisements
Advertisements
प्रश्न
What are esters? How are esters prepared? Write the chemical equation for the reaction involved. What happens when an ester reacts with sodium hydroxide? Write the chemical equation for the reaction and also state the name and use of this reaction.
उत्तर
Esters
They are sweet-smelling organic compounds, with the general formula RCOOR' (R and R' are similar or dissimilar alkyl groups).
Preparation of ester
Esters are prepared by esterification which is a reaction between carboxylic acids and alcohols in the presence of an acid.
For example, when ethanoic acid reacts with ethanol in the presence of an acid, ethyl ethanoate is formed. Ethyl ethanoate is an ester, and has a sweet smell.
\[{{CH}_3 {CH}_2 OH}_{\text{Ethanol}} + {{CH}_3 COOH}_{\text{Ethanoic acid}} \to^{\text{Acid}} {{CH}_3 {COOCH}_2 {CH}_3}_{\text{Ethyl ethanoate }(\text{Ester})} + {H_2 O}_{\text{Water}}\]
Reaction with sodium hydroxide
Ethyl acetate (an ester) reacts with sodium hydroxide (base) to form ethanol (alcohol) and ethanoic acid (carboxylic acid).
\[{{CH}_3 {COOCH}_2 {CH}_3}_{\text{Ethyl ethanoate }(\text{Ester})} \to^{NaOH} {{CH}_3 {CH}_2 OH}_{\text{Ethanol}} + {{CH}_3 COOH}_{\text{Ethanoic acid}}\]
Thus, alcohol and carboxylic acid can be reclaimed from an ester by reacting it with an acid or a base. This reaction is used in the preparation of soaps and it is known as saponification reaction.
APPEARS IN
संबंधित प्रश्न
From which plant is litmus paper or litmus solution obtained?
- Moss
- Rose
- Hibiscus
- Lichen
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
Name one natural source of the following acids:
Lactic acid
Write a balanced chemical equation of the reaction which takes place.
Wasp stings can be treated with:
(a) baking soda
(b) vinegar
(c) washing soda
(d) milk of magnesia
Write the chemical formula of sodium carbonate decahydrate.
Name the product formed when Cl2 and H2 produced during the electrolysis of brine are made to combine.
Explain the following:
Lead carbonate does not react with dilute HCl.
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
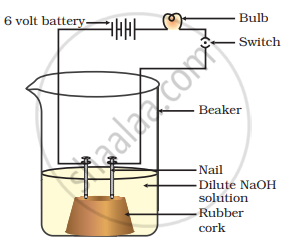
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
What property do acids and bases have in common? Explain it with an example.
