Advertisements
Advertisements
Question
What are esters? How are esters prepared? Write the chemical equation for the reaction involved. What happens when an ester reacts with sodium hydroxide? Write the chemical equation for the reaction and also state the name and use of this reaction.
Solution
Esters
They are sweet-smelling organic compounds, with the general formula RCOOR' (R and R' are similar or dissimilar alkyl groups).
Preparation of ester
Esters are prepared by esterification which is a reaction between carboxylic acids and alcohols in the presence of an acid.
For example, when ethanoic acid reacts with ethanol in the presence of an acid, ethyl ethanoate is formed. Ethyl ethanoate is an ester, and has a sweet smell.
\[{{CH}_3 {CH}_2 OH}_{\text{Ethanol}} + {{CH}_3 COOH}_{\text{Ethanoic acid}} \to^{\text{Acid}} {{CH}_3 {COOCH}_2 {CH}_3}_{\text{Ethyl ethanoate }(\text{Ester})} + {H_2 O}_{\text{Water}}\]
Reaction with sodium hydroxide
Ethyl acetate (an ester) reacts with sodium hydroxide (base) to form ethanol (alcohol) and ethanoic acid (carboxylic acid).
\[{{CH}_3 {COOCH}_2 {CH}_3}_{\text{Ethyl ethanoate }(\text{Ester})} \to^{NaOH} {{CH}_3 {CH}_2 OH}_{\text{Ethanol}} + {{CH}_3 COOH}_{\text{Ethanoic acid}}\]
Thus, alcohol and carboxylic acid can be reclaimed from an ester by reacting it with an acid or a base. This reaction is used in the preparation of soaps and it is known as saponification reaction.
APPEARS IN
RELATED QUESTIONS
Why should curd and sour substances not be kept in brass and copper vessels?
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
A solution reacts with marble chips to produce a gas which turns lime water milky. The solution contains:
(a) Na2SO4
(b) CaSO4
(c) H2SO4
(d) K2SO4
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
Predict the new compound formed which imparts a blue-green colour to solution.
What ions are present in the solutions of following substances? (write the symbols only)
Nitric acid
What ions are present in the solutions of following substances? (write the symbols only)
Magnesium hydroxide
What is the chemical name of bleaching powder?
Read the following statements:
I. When a red litmus paper is dipped into reaction mixture of a saponification reaction, it turns blue and the reaction is exothermic.
II. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is exothermic.
III. When a red litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
IV. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
Which of the above statements are correct:
(A) I, and II
(B) II and III
(C) III and IV
(D) I and IV
Common salt besides being used in kitchen can also be used as the raw material for making
- washing soda
- bleaching powder
- baking soda
- slaked lime
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
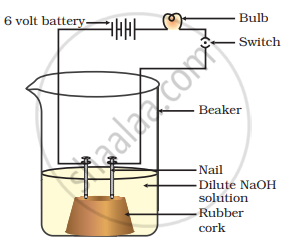
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
