Advertisements
Advertisements
Question
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
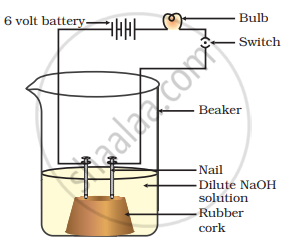
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Options
(i) and (iii)
(ii) and (iv)
(ii) only
(iv) only
Solution
(ii) only
Explanation -
Bulb will glow because the acid will furnish ions which conduct electricity.
APPEARS IN
RELATED QUESTIONS
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
Fill in the blank in the following sentences:
Substances do not show their acidic properties without.......................... .
Name the gas evolved when zinc granules are treated/heated with:
hydrochloric acid solution
What would be the colour of litmus in a solution of sodium carbonate?
Explain why, sodium hydrogencarbonate is used as an antacid.
Which of the following gives the correct increasing order of acidic strength?
Which of the following are present in a dilute aqueous solution of hydrochloric acid?
______ & ______ metals do not react with HCl or HNO3.
The taste of acid is ______.
The metal that will not produce hydrogen gas when reacted with dilute acids.
