Advertisements
Advertisements
Question
Read the following statements:
I. When a red litmus paper is dipped into reaction mixture of a saponification reaction, it turns blue and the reaction is exothermic.
II. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is exothermic.
III. When a red litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
IV. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
Which of the above statements are correct:
(A) I, and II
(B) II and III
(C) III and IV
(D) I and IV
Solution
(A)
The saponification reaction is exothermic and the soap solution is basic in nature so it turns red litmus paper into blue. When a blue litmus paper is dipped, its colour does not change.
APPEARS IN
RELATED QUESTIONS
Define an acid and a base. Give two examples of each.
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Name X and Y
What is a neutralization reaction? Give an example.
A solution of NaCl
(i) will turn red litmus blue
(ii) will turn pH paper green
(iii) will turn blue litmus red
(iv) will not affect litmus
Egg shell is made up of ____________.
Which of the following gives the correct increasing order of acidic strength?
To protect tooth decay, we are advised to brush our teeth regularly. The nature of the tooth paste commonly used is
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
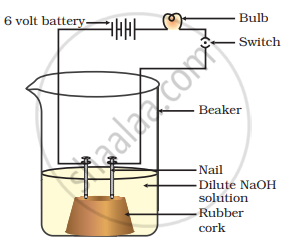
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made?

- In place of zinc granules, same amount of zinc dust is taken in the test tube
- Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
- In place of zinc, copper turnings are taken
- Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
Solutions of acids conduct ______.
