Advertisements
Advertisements
प्रश्न
Read the following statements:
I. When a red litmus paper is dipped into reaction mixture of a saponification reaction, it turns blue and the reaction is exothermic.
II. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is exothermic.
III. When a red litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
IV. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
Which of the above statements are correct:
(A) I, and II
(B) II and III
(C) III and IV
(D) I and IV
उत्तर
(A)
The saponification reaction is exothermic and the soap solution is basic in nature so it turns red litmus paper into blue. When a blue litmus paper is dipped, its colour does not change.
APPEARS IN
संबंधित प्रश्न
How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously, and why?
Fill in the blank in the following sentences:
Substances do not show their acidic properties without.......................... .
State the common and chemical names of the compound formed when plaster of Paris is mixed with water.
Explain why, sodium hydrogencarbonate is used as an antacid.
State two uses each of the following compounds:
Sodium hydroxide
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
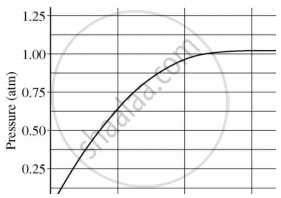
Which of the following statements is true for acids?
What happens when nitric acid is added to egg shell?
______ & ______ metals do not react with HCl or HNO3.
