Advertisements
Advertisements
Question
Define an acid and a base. Give two examples of each.
Solution
Acids: Substances that ionise on dissolving in water producing hydrogen ions (H+ions) are called acids. Acids are sour in taste and they turn blue litmus to red.
Examples: Hydrochloric acid, HCl, and sulphuric acid, H2SO4.
Bases: Substances that ionise on dissolving in water producing hydroxide ions (OH- ions) are called bases. Bases are bitter in taste, slippery to touch and they turn red litmus to blue.
Examples: Magnesium hydroxide, Mg(OH)2, and sodium hydroxide, NaOH.
APPEARS IN
RELATED QUESTIONS
Why do HCl, HNO3, etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
With the help of labelled diagrams, describe an activity to show that acids produce ions only in aqueous solutions.
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
Complete and balance the following chemical equations:
`Ca(OH)_2 + Cl_2 ->`
What is the chemical formula of bleaching powder?
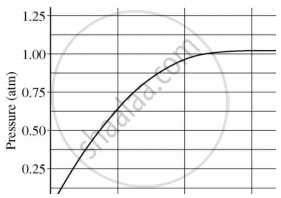
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
Common salt besides being used in kitchen can also be used as the raw material for making
- washing soda
- bleaching powder
- baking soda
- slaked lime
Vinegar is ______ in taste.
Can we taste acids and bases to identify them?
What is the correct procedure for mixing acid and water? Why?
