Advertisements
Online Mock Tests
Chapters
![Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 chapter 2 - Acids, Bases and Salts Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 chapter 2 - Acids, Bases and Salts - Shaalaa.com](/images/9352831810-chemistry-science-english-class-10_6:7eeaf5db0f4a49c580d3c8746ff5ab36.jpg)
Advertisements
Solutions for Chapter 2: Acids, Bases and Salts
Below listed, you can find solutions for Chapter 2 of CBSE Lakhmir Singh for Chemistry (Science) [English] Class 10.
Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 2 Acids, Bases and Salts Exercise 1 [Pages 66 - 69]
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
(a) methyl orange
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
litmus
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
red cabbage extract
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
litmus
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Name an indicator which is red in acid solution but turns blue in basic solution.
Name an indicator which is pink in alkaline solution but turns colourless in acidic solution.
When a solution is added to a cloth strip treated with onion extract, then the smell of onion cannot be detected. State whether the given solution contains an acid or a base.\
When a solution is added to vanilla extract, then the characteristic smell of vanilla cannot be detected. State whether the given solution is an acid or a base.
How will you test for the gas which is liberated when hydrochloric acid reacts with an active metal?
Name the gas evolved when dilute HCl reacts with sodium hydrogencarbonate. How is it recognised?
Give the names and formulae of two strong acids and two weak acids.
Name one natural source of the following acids:
Citric acid
Name one natural source of the following acids:
Oxalic acid
Name one natural source of the following acids:
Lactic acid
Name one natural source of the following acids:
(d) Tartaric acid
Name one animal and one plant whose stings contain formic acid (or methanoic acid).
How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with zinc granules.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with aluminium powder.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with iron filings.
Complete and balance the following chemical equations:
Zn (s) + HCI (aq) →
Complete and balance the following chemical equations
Na2 CO3 (s) + HCI (aq) →
Complete and balance the followingchemicalequations:
NaHCO3 (S) + HCI (aq) →
Complete and balance the following chemical equations:
NaOH (aq) HCI (aq)→
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
Fill in the blank in the following sentences:
Acids have a ..........................taste and they turn..........................litmus to..........................
Fill in the blank in the following sentences:
Substances do not show their acidic properties without.......................... .
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
Fill in the blank in the following sentences:
Those substances whose smell (or odour) changes in acidic or basic solution are called .......................... indicators.
Fill in the blank in the following sentences:
Onion and vanilla extract are..........................indicators.
What is an indicator? Name three common indicators.
Name the acid-base indicator extracted from lichen.
What colour does the turmeric paper turn when put in an alkaline solution?
What is an olfactory indicator?
What is the effect of adding sodium hydroxide solution to these olfactory indicators?
Name two olfactory indicators?
Give chemical equation of the reaction involved.
What happens when an acid reacts with a metal?
Which gas is usually liberated when an acid reacts with a metal?
How will you test for the presence of this gas?
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
What happens when an acid reacts with a metal hydrogencarbonate? Write equation of the reaction which takes place.
What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced chemical equation of the reaction involved.
How will you test for the presence of this gas?
Which gas is liberated when dilute hydrochloric acid reacts with sodium carbonate?
What happens when an acid reacts with a base?
Explain by taking the example of hydrochloric acid and sodium hydroxide. Give equation of the chemical reaction which takes place. What is the special name of such a reaction?
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
What are organic acids and mineral acids?
Give two examples each of organic acids and mineral acids.
State some of the uses of mineral acids in industry.
What is meant by strong acids and weak acids? Classify the following into strong acids and weak acids:
HCI, CH3 COOH, H2 SO 4, HNO3, H2 CO3 , H2 SO3
Why do HCl, H2SO4, HNO3, etc., show acid character in aqueous solutions while solutions of compounds like C6H12O6 (glucose) and C2H5OH (alcohol) do not show acidic character?
Explain with an example. Give the chemical equation of the reaction which takes place.
What is a neutralisation reaction?
Why should curd and other sour foodstuffs (like lemon juice, etc.) not be kept in metal containers (such as copper and brass vessels)?
What is produced if an acid is added to a base?
Dry HCl gas does not change the colour of dry blue litmus paper. Why?
What colour does phenolphthalein indicator turn when added to an alkali (such as sodium hydroxide)?
Why do acids not show acidic behaviour in the absence of water?
Why does an aqueous solution of an acid conduct electricity?
Why does distilled water not conduct electricity, whereas rain water does?
What happens when an acid reacts with a metal carbonate? Explain with the help of an example. Write chemical equation of the reaction involved.
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
What happens when carbon dioxide gas is passed through lime water for a short time?
Write equations of the reactions involved.
With the help of labelled diagrams, describe an activity to show that acids produce ions only in aqueous solutions.
Which element is common to all acids?
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Which of the following types of medicine is used for treating indigestion caused by over-eating?
(a) antibiotic
(b) analgesic
(c) antacid
(d) antiseptic
A solution reacts with marble chips to produce a gas which turns lime water milky. The solution contains:
(a) Na2SO4
(b) CaSO4
(c) H2SO4
(d) K2SO4
One of the following is not an organic acid. This is:
(a) ethanoic acid
(b) formic acid
(c) citric acid
(d) carbonic acid
The property which is not shown by acids is:
(a) they have sour taste
(b) they feel soapy
(c) they turn litmus red
(d) their pH is less than seven
The indicators which turn red in acid solution are:
(a) turmeric and litmus
(b) phenolphthalein and methyl orange
(c) litmus and methyl orange
(d) phenolphthalein and litmus
The discomfort caused by indigestion due to overeating can be cured by taking:
Vinegar
Lemon juice
Baking soda
Caustic soda
The property which is common between vinegar and curd is that they:
(a) have sweet taste
(b) have bitter taste
(c) are tasteless
(d) have sour taste
The indicator which produces a pink colour in an alkaline solution is:
(a) methyl orange
(b) turmeric paper
(c) phenolphthalein
(d) litmus paper
A solution reacts with zinc granules to give a gas which burns with a 'pop' sound. The solution contains:
(a) Mg(OH)2
(b) Na2CO3
(c) NaCl
(d) HCl
When a piece of limestone reacts with dilute HCl, a gas X is produced. When gas X is passed through lime water then a white precipitate Y is formed. On passing excess of gas X, the white precipitate dissolves forming a soluble compound Z.
- What are X, Y and Z?
- Write equations for the reactions which take place:
- When limestone reacts with dilute HCl.
- When gas X reacts with lime water to form white precipitate Y.
- When excess of gas X dissolves white precipitate Y to form a soluble compound Z.
If someone is suffering from the problem of acidity after overeating, which of the following would you suggest as remedy?
Lemon juice, Vinegar, Banking soda solution
Give reason for your choice.
Write a balanced chemical equation of the reaction which takes place.
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
Predict the new compound formed which imparts a blue-green colour to solution.
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
Name the natural indicator present in curry stain.
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
Explain the changes in colour of this indicator which take place during washing and rinsing the shirt.
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
What is the nature of soap (acidic/basic) as shown by the indicator present in curry stain?
You have been provided with three test-bubes. One of these test-tubes contains distilled water and the other two contain an acidic and a basic solution respectively. If you are given only blue litmus paper, how will you identify the contents of each test-tube?
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
How is the neutralisation of a carbonate with an acid different from the neutralisation of an oxide or a hydroxide?
What happens to the H+ ions, of the solution, when an acid is neutralised?
What happens to temperature of the solution, when an acid is neutralised?
Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 2 Acids, Bases and Salts Exercise 2 [Pages 79 - 83]
Name the gas evolved when zinc granules are treated/heated with:
hydrochloric acid solution
Name the gas evolved when zinc granules are treated/heated with:
sodium hydroxide solution
What is the common name of water soluble bases?
What is common in all the water soluble bases (or alkalis)?
Why does tooth decay start when the pH of mouth is lower than 5.5?
What is the pH of a neutral solution?
Which is more acidic : a solution of pH = 2 or a solution of pH = 6?
hich is more basic (or more alkaline) : a solution of pH = 8 or a solution of pH = 11?
Name the scientist who developed the pH scale.
Name the indicator which can give us an idea of how strong or weak an acid or base is.
The pH of soil A is 7.5 while that of soil B is 4.5. Which of the two soils, A or B, should be treated with powdered chalk to adjust its pH and why?
What is the name of the indicator which can be used for testing the pH of a solution?
What colour will universal indicator show if you add it to the following substances?
(a) potassium hydroxide, pH = 12
(b) soda water, pH = 5
(c) sulphuric acid, pH = 2
A beaker of concentrated hydrochloric acid has a pH of 1. What colour will full range universal indicator turn if it is added to this beaker? Is it a strong or a weak acid?
Two solutions X and Y are tested with universal indicator. Solution X turns orange whereas solution Y turns red. Which of the solutions is a stronger acid?
Two solutions A and B have pH values of 3.0 and 9.5 respectively. Which of these will turn litmus solution from blue to red and which will turn phenolphthalein from colourless to pink?
Two drinks P and Q gave acidic and alkaline reactions, respectively. One has a pH value of 9 and the other has a pH value of 3. Which drink has the pH value of 9?
Two solutions X and Y have pH = 4 and pH = 8, respectively. Which solution will give alkaline reaction and which one acidic?
Fill in the following blank with suitable words:
Acids have a pH...........than 7.
Fill in the following blank with suitable words:
Alkalis have a pH.............. than 7.
Fill in the following blank with suitable words:
Neutral substances have a pH of ................
Fill in the following blank with suitable words:
The more acidic a solution, the ................. the pH.
Fill in the following blank with suitable words:
The more alkaline a solution, the............... the pH.
Fresh milk has a pH of 6. When it changes into curd (yogurt), will its pH value increase or decrease? Why?
What is a universal indicator? For what purpose is it used?
How does a universal indicator work?
Water is a neutral substance. What colour will you get when you add a few drops of universal indicator to a test-tube containing water?
Which chemical is injected into the skin of a person during an ant's sting?
How can the effect of these stings be neutralised?
Which chemical is injected into the skin of a person during the nettle leaf hair sting?
How can the effect of these stings be neutralised?
Explain the pH change as the cause of tooth decay. How can tooth decay caused by pH change be prevented?
Explain how pH change in the lake water can endanger the lives of aquatic animals (like fish). What can be done to lessen the danger to the lives of aquatic animals in the lake?
What happens during a bee sting? What is its remedy?
What happens during a wasp sting? What is its remedy?
Why is it wrong to treat a bee sting with vinegar?
Why is it wrong to treat a wasp sting with baking soda solution?
What does the pH of a solution signify? Three solution A, B and C have pH values of 6, 4 and 10 respectively. Which of the solutions is highly acidic?
A farmer has found that the pH of soil in his fields is 4.2. Name any two chemical materials which he can mix with the soil to adjust its pH.
The pH values of six solutions A to F are given below:
A = 0, B = 11, C = 6, D = 3, E = 13, F = 8
Which of the above solutions are (i) acids (ii) alkalis?
(b) Name the acid or alkalis used to make (i) car batteries (ii) explosives (iii) soaps (iv) fertilisers.
The pH of a cold drink is 5. What will be its action on blue and red litmus solutions?
The pH values of three acids A, B and C having equal molar concentrations are 5.0, 2.8 and 3.5 respectively. Arrange these acids in order of the increasing acid strengths.
Under what soil conditions do you think a farmer would treat the soil of his fields with quicklime (calcium oxide), or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Which acid is produced in our stomach? What happens if there is an excess of acid in the stomach? How can its effect be cured?
The soil in a field in highly acidic. Name two materials which can be added to this soil to reduce its acidity. Give the reason for your choice.
What is meant by strong bases and weak bases? Classify the following into strong bases and weak bases:
NH4OH, Ca(OH)2, NaOH, KOH, Mg(OH)2
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
What ions are present in the solutions of following substances? (write the symbols only)
Nitric acid
What ions are present in the solutions of following substances? (write the symbols only)
Sulphuric acid
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
What ions are present in the solutions of following substances? (write the symbols only)
Potassium hydroxide
What ions are present in the solutions of following substances? (write the symbols only)
Magnesium hydroxide
What would you expect the pH of pure water to be?
What colour would the universal indicator show in an aqueous solution of sugar? Why?
A sample of rain water turned universal indicator paper yellow. What would you expect it pH to be? Is it a strong or a weak acid?
Name two common antacids.
What do you think will be the pH in the stomach of a person suffering from indigestion: less than 7 or more than 7?
What do you think will be the pH of an antacid solution: less than 7 or more than 7?
How does an antacid work?
Name two common antacids.
Separate the following into substances having pH values above and below 7. How do these influence litmus paper?
(i) Lemon juice
(ii) Solution of washing soda
(iii) Toothpaste
(iv) Vinegar
(v) Stomach juices
Do basic solutions also have H+ (aq) ions? If yes, then why are these basic?
When a solution becomes more acidic, does the pH get higher or lower?
Define an acid and a base. Give two examples of each.
Give the names and formulae of two strong bases and two weak bases.
What type of ions is formed:
when a base (or alkali) is dissolved in water?
What type of ions is formed when an acid is dissolved in water?
Write the neutralisation reaction between acids and bases in terms of the ions involved.
Write any two important uses of bases.
Name the gas evolved when zinc granules are treated/heated with:
sodium hydroxide solution
What happens when bases react with non-metal oxides? Explain with the help of an example. What does this reaction tell us about the nature of non-metal oxides?
What effect does the concentration of H+ (aq) ions have on the nature of a solution?
What effect does the concentration of OH− ions have on the nature of a solution?
Someone put some universal indicator paper into vinegar. The pH is 3. What does this tell you about the vinegar?
Someone put some universal indicator paper onto wet soap. The pH is 8. What does this tell you about the soap?
State whether a solution is acidic, alkaline or neutral if its pH is:
(i) 9
(ii) 4
(iii) 7
(iv) 1
(v) 10
(vi) 3
One of the following is a medicine for indigestion. This is:
(a) sodium hydroxide
(b) manganese hydroxide
(c) magnesium hydroxide
(d) potassium hydroxide
Bee sting contains:
(a) an acidic liquid
(b) a salt solution
(c) an alkaline liquid
(d) an alcohol
Wasp sting contains:
(a) a sugar solution
(b) an acidic liquid
(c) a salt solution
(d) an alkaline liquid
One of the following does not inject an acidic liquid into the skin through its sting. This is:
(a) honey bee
(b) ant
(c) wasp
(d) nettle leaf hair
A solution turns red litmus blue, its pH is likely to be ______.
1
4
5
10
A solution turns blue litmus red. Its pH is likely to be:
(a) 7
(b) 5
(c) 8
(d) 14
A solution turns phenolphthalein indicator pink. The most likely pH of this solution will be:
(a) 6
(b) 4
(c) 9
(d) 7
The colour of methyl orange indicator in a solution is yellow. The pH of this solution is likely to be:
(a) 7
(b) less than 7
(c) 0
(d) more than 7
Bee stings can be treated with:
(a) vinegar
(b) sodium hydrogencarbonate
(c) potassium hydroxide
(d) lemon juice
Wasp stings can be treated with:
(a) baking soda
(b) vinegar
(c) washing soda
(d) milk of magnesia
It has been found that rubbing vinegar on the stung area of the skin of a person gives him relief. The person has been stung by:
(a) wasp
(b) ant
(c) honey bee
(d) nettle leaf hair
Fresh milk has a pH of 6. When milk changes into curd, the pH value will:
(a) become 7
(b) become less than 6
(c) become more than 7
(d) remain unchanged
The acid produced naturally in our stomach is:
(a) acetic acid
(b) citric acid
(c) hydrochloric acid
(d) sulphuric acid
The daffodil plants grow best in a soil having a pH range of 6.0 to 6.5. If the soil in a garden has a pH of 4.5, which substance needs to be added to the soil in order to grow daffodils?
(a) salt
(b) lime
(c) sand
(d) compost
A milkman adds a very small amount of baking soda to fresh milk. Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
Which of the following elements would from oxides which would indicate pH values less than seven, using moist pH paper?
Magnesium, Carbon, Sulphur, Hydrogen, Copper
The pH values of five solutions A, B, C, D and E are given below:
| A | 1 |
| B | 5 |
| C | 7 |
| D | 11 |
| E | 13 |
Which solution is weakly alkaline.
The pH values of five solutions A, B, C, D and E are given below:
| A | 1 |
| B | 5 |
| C | 7 |
| D | 11 |
| E | 13 |
Which solution isneutral neutral.
The pH values of five solutions A, B, C, D and E are given below:
| A | 1 |
| B | 5 |
| C | 7 |
| D | 11 |
| E | 13 |
The pH values of five solutions A, B, C, D and E are given below:
| A | 1 |
| B | 5 |
| C | 7 |
| D | 11 |
| E | 13 |
The pH values of five solutions A, B, C, D and E are given below:
| A | 1 |
| B | 5 |
| C | 7 |
| D | 11 |
| E | 13 |
Here are some results of solutions tested with universal indicator paper:
| Sulphuric acid | : Red |
| Metal polish | : Dark blue |
| Washing-up liquid | : Yellow |
| Milk of magnesia | : Light blue |
| Oven cleaner | : Purple |
| Car batter acid | : Pink |
Arrange the solutions in order of their increasing pH value (starting with the one with the lowest pH).
Solution A turns universal indicator blue to purple whereas solution B turns universal indicator orange to red.
What will be the action of solution A on litmus?
Solution A turns universal indicator blue to purple whereas solution B turns universal indicator orange to red
What will be action of solution B on litmus?
Solution A turns universal indicator blue to purple whereas solution B turns universal indicator orange to red.
Name any two substances which can give solutions like A.
Solution A turns universal indicator blue to purple whereas solution B turns universal indicator orange to red.
Name any two substances which can give solutions like B.
Solution A turns universal indicator blue to purple whereas solution B turns universal indicator orange to red.
What sort of reaction takes place when solution A reacts with solution B?
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
bee stings?
How could the person change his eating habits to lessen chances of suffering from tooth decay?
Explain why the pH in a person's mouth becomes lower after each meal.
What damage could be caused while the pH is low?
A group of students measured the pH of some substances they found in their homes. Their results are given in the following table:
| Substance | pH | Substance | pH |
| Apples | 3.0 | Salt | 7.0 |
| Baking soda | 8.5 | Sugar | 7.0 |
| Black coffee | 5.0 | Toothpaste | 9.0 |
| Household ammonia | 12.0 | Vinegar | 3.0 |
| Lemon juice | 2.5 | Washing soda | 11.5 |
| Milk | 6.5 |
(a) What would the students have used to measure the pH?
(b) Which solution is the most acidic?
(c) Which solution is the most alkaline?
(d) Which solutions are neutral?
(e) Which solution can be used to treat wasp stings?
(f) Which solutions can be used to treat bee stings?
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Name X and Y
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 2 Acids, Bases and Salts Exercise 3 [Pages 96 - 100]
What is the chemical formula of washing soda?
What is the chemical formula of baking soda?
Write the chemical formula of soda ash?
Write the chemical formula of sodium carbonate decahydrate.
State whether the following statement is true or false:
Copper sulphate crystals are always wet due to the presence of water of crystallisation in them.
Which of the following salt has a blue colour and why?
CuSO4.5H2O or CuSO4
What would be the colour of litmus in a solution of sodium carbonate?
State the common and chemical names of the compound formed when plaster of Paris is mixed with water.
With which substance should chlorine be treated to get bleaching powder?
What is the commercial name of calcium sulphate hemihydrate?
Name the product formed when Cl2 and H2 produced during the electrolysis of brine are made to combine.
Name a calcium compound which hardens on wetting with water.
Name a sodium compound which is a constituent of many dry soap powders.
Name a metal carbonate which is soluble in water.
Name an acid which is present in baking powder.
Name the metal whose carbonate is known as washing soda.
Which compound is used as an antacid in medicine : NaHCO3 or Na2CO3?
What is the common name of (a) NaHCO3 and (b) Na2CO3.10H2O?
Write the chemical name and formula of common salt.
Write the chemical name and formula of caustic soda.
What are the two main ways in which common salt (sodium chloride) occurs in nature?
Name the major salt present in sea-water.
How is common salt obtained from sea-water?
Why is sodium chloride required in our body?
Name three chemicals made from common salt (or sodium chloride).
Give any two uses of common salt (sodium chloride).
What name is given to the common salt which is mined from underground deposits? How was this salt formed?
Name the salt which is used as preservative in pickles, and in curing meat and fish.
Name the raw material used for the production of caustic soda.
The electrolysis of an aqueous solution of sodium chloride gives us three products. Name them.
During the electrolysis of a saturated solution of sodium chloride, where is:
chlorine formed?
During the electrolysis of a saturated solution of sodium chloride, where is:
hydrogen formed?
During the electrolysis of a saturated solution of sodium chloride, where is:
sodium hydroxide formed?
Fill in the following blank:
Common salt is obtained from sea-water by the process of.............
Fill in the following blank:
Rock salt is mined just like ......................
Fill in the following blank:
Chemical formula of washing soda is ..................
Fill in the following blank:
Sodium hydrogen carbonate is .................. soda whereas sodium carbonate is ................. soda.
Fill in the following blank:
The chemical formula of plaster of Paris is ............
Complete and balance the following chemical equations:
`NaCI (aq) + H_2O (l) `![]()
Complete and balance the following chemical equations:
`NaHCO_3` 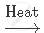
Complete and balance the following chemical equations:
`NaCl + NH_3 + H_2O +CO_2`→
Complete and balance the following chemical equations:
`Ca(OH)_2 + Cl_2 ->`
What is washing soda?
State two properties and two uses of washing soda.
Write the formulae of sodium chloride and sodium carbonate.
Explain why an aqueous solution of sodium chloride is neutral but an aqueous solution of sodium carbonate is basic (or alkaline). Write chemical equations of the reactions involved.
Write the chemical formula of ammonium chloride. Explain why an aqueous solution of ammonium chloride is acidic in nature? Illustrate your answer with the help of a chemical equation.
What is baking soda?
Write the chemical name of baking soda. Give the important uses of baking soda. How does baking soda differ chemically from washing soda?
Describe how sodium hydrogencarbonate (baking soda) is produced on a large scale. Write equation of the reaction involved.
What happens when a cold and concentrated solution of sodium chloride reacts with ammonia and carbon dioxide? Write the chemical equation of the reaction which takes place.
What is meant by "water of crystallisation" in a substance? Explain with an example.
How would you show that blue copper sulphate crystals contain water of crystallisation?
Explain how anhydrous copper sulphate can be used to detect the presence of moisture (water) in a liquid
What is the common name of sodium hydrogencarbonate?
What happens when a solution of sodium hydrogencarbonate is heated? Write equation of the reaction involved.
Explain why, sodium hydrogencarbonate is used as an antacid.
What will happen if heating is not controlled while preparing plaster of Paris?
Write an equation to show the reaction between Plaster of Paris and water.
What happens when copper sulphate crystals are heated strongly? Explain with the help of an equation.
What happens when a few drops of water are added to anhydrous copper sulphate? Explain with the help of an equation.
Name two constituents of baking powder.
How does baking powder differ from baking soda?
Explain the action of baking powder in the making of cake (or bread). Write equation of the reaction involved.
What is the chemical name of bleaching powder?
What is the chemical formula of bleaching powder?
What are the materials used for the preparation of bleaching powder?
State one use of bleaching powder (other than bleaching).
What does a soda-acid type fire extinguisher contain? How does it work? Explain the working of a soda-acid fire extinguisher with the help of a labelled diagram.
Name the sodium compound which is used for softening hard water.
Which compound of calcium is used for disinfecting drinking water supply?
Name a metal compound which has detergent properties (cleansing properties).
Name one compound of calcium which is used for removing the colour of a coloured cloth.
Plaster of Paris has a remarkable property of setting into a hard mass on addition of water.
Name the substance obtained by the action of chlorine on solid (dry) slaked lime.
What is gypsum? What happens when gypsum is heated to 100°C (373 K)?
Name a sodium compound which is used for making borax and glass.
Name the compound which is used in hospitals for setting fractured bones.
Which is the real bleaching agent present in bleaching powder?
What is "baking powder"? How does it make the cake soft and spongy?
In addition to sodium hydrogencarbonate, baking powders contain a substance X. Name the substance X. What is the role of substance X in the baking powder?
State two uses each of the following compounds:
Sodium hydroxide
State two uses each of the following compounds:
Chlorine
State two uses each of the following compounds:
Hydrogen
State two uses each of the following compounds:
Hydrochloric acid
What is the common name of the compound CaOCl2?
Name the raw material used for the preparation of plaster of Paris.
Which property of plaster of Paris is utilised in making casts for broken limbs in hospitals?
Explain why chlorine is used for sterilising drinking water supply.
What happens when a concentrated solution of sodium chloride (brine) is electrolysed? Write the equation of the reaction involved.
Why is the electrolysis of a concentrated solution of sodium chloride known as chlor-alkali process?
Name three products of the chlor-alkali process. State two uses of each of these products.
Describe how washing soda is produced starting from sodium chloride (common salt). Write equations of all the reactions involved.
State whether an aqueous solution of washing soda is acidic or alkaline? Give reason for your answer.
What is meant by saying that washing soda has detergent properties?
Give two important uses of washing soda.
How is bleaching powder prepared? Write chemical equation of the reaction involved in the preparation of bleaching powder.
What is bleaching powder?
What happens when bleaching powder reacts with dilute sulphuric acid? Give equation of the reaction involved.
State two important uses of bleaching powder.
What is plaster of Paris? Write the chemical formula of plaster of Paris.
How is plaster of Parish prepared? Write chemical equation of the reaction involved.
Plaster of Paris should be stored in a moisture-proof container. Explain why?
State two important use of plaster of Paris.
What is a salt?
Give the names and formulae of any two salts. Also name the acids and bases from which these salt may be obtained.
What is meant by 'a family of salts'? Explain with examples.
What is meant by 'hydrated' and 'anhydrous' salts? Explain with examples.
Write the names, formulae and colours of any two hydrated salts.
What will be the colour of litmus in an aqueous solution of ammonium chloride salt?
The salt which will give an acidic solution on dissolving in water is:
(a) KCl
(b) NH4Cl
(c) Na2Co3
(d) CH3COONa
One of the following salts will give an alkaline solution on dissolving in water. This is:
a) Na2CO3
(b) Na2SO4
(c) NaCl
(d) (NH4)2 SO4
The salt which will give a neutral solution on dissolving in water will be:
(a) CH3COONa
(b) NH4Cl
(c) KCl
(d) Na2CO3
The products of chlor-alkali process are:
(a) NaCl, Cl2 and H2
(b) H2, Cl2 and NaOH
(c) Cl2, Na2CO3 and H2O
(d) NaOH, Cl2 and HCl
The number of molecules of water of crystallisation present in washing soda crystals is:
(a) five
(b) two
(c) ten
(d) seven
The salt whose aqueous solution will turn blue litmus to red is:
(a) ammonium sulphate
(b) sodium acetate
(c) sodium chloride
(d) potassium carbonate
The aqueous solution of one of the following salts will turn red litmus to blue. This salt is:
(a) potassium sulphate
(b) sodium sulphate
(c) sodium chloride
(d) potassium carbonate
The salt whose aqueous solution will have no effect on either red litmus or blue litmus is
(a) potassium sulphate
(b) sodium carbonate
(c) ammonium sulphate
(d) sodium acetate
The aqueous solution of one of the following salts will turn phenolphthalein indicator pink. This salt is:
(a) KCl
(b) K2SO4
(c) K2CO3
(d) KNO3
The formula of baking soda is:
(a) K2CO3
(b) KHCO3
(c) NaHCO3
(d) Na2CO3
Which of the following is treated with chlorine to obtain bleaching powder?
(a) CaSO4
(b) Ca(OH)2
(c) Mg(OH)2
(d) KOH
Plaster of Paris is prepared by heating one of the following to a temperature of 100°C. This is:
(a) CaSO3.2H2O
(b) CaCl2.2H2O
(c) CaCO3.2H2O
(d) CaSO4.2H2O
A salt whose aqueous solution will have a pH of more than 7 will be:
(a) K2CO3
(b) K2SO4
(c) NaCl
(d) NH4Cl
A salt is dissolved in water and the pH of this salt solution is measured with a universal indicator paper. If the pH of solution is less than 7, the salt is most likely to be:
(a) CH3COONa
(b) Na2CO3
(c) KCl
(d) NH4Cl
Which of the following salts will give an aqueous solution having pH of almost 7?
(a) NH4NO3
(b) NH4Cl
(c) CaCl2
(d) KCl
P and Q are aqueous solutions of sodium chloride and sodium hydroxide, respectively. Which of these will turn:
(a) blue litmus red?
P and Q are aqueous solutions of sodium chloride and sodium hydroxide, respectively. Which of these will turn:
red litmus blue?
The metal salt A is blue in colour. When salt A is heated strongly over a burner, then a substance B is eliminated and a white powder C is left behind. When a few drops of a liquid D are added to powder C, it becomes blue again. What could be A, B, C and D
When the concentrated aqueous solution of substance X is electrolysed, then NaOH, Cl2 and H2 are produced. Name the substance X. What is the special name of this process?
Consider the following substance:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which two substance combine to form bleaching powder?
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which four substances are utilised in the production of washing soda?
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound represents plaster of Paris?
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound is a part of baking powder?
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound is used as an antacid?
Give one example of a salt which gives an aqueous solution having:
pH less than 7
Give one example of a salt which gives an aqueous solution having:
pH equal to 7
Give one example of a salt which gives an aqueous solution having:
pH more than 7
A compound X which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water.
Identify the compound X
A compound X which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water.
Write the chemical equation for its preparation
A compound X which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water.
For what purpose is it used in hospitals?
Consider the following salts:
Na2CO3, NaCl, NH4Cl, CH3COONa, K2SO4, (NH4)2SO4
Which of salts will give:
acidic solutions?
Consider the following salts:
Na2CO3, NaCl, NH4Cl, CH3COONa, K2SO4, (NH4)2SO4
Which of salts will give:
neutral solutions?
Consider the following salts:
Na2CO3, NaCl, NH4Cl, CH3COONa, K2SO4, (NH4)2SO4
Which of these salts will give:
basic solutions (or alkaline solutions)?
A white powdery substance having strong smell of chlorine is used for disinfecting drinking water supply at waterworks. Identify the substance. Give its chemical name and write the chemical reaction for its preparation.
A salt X when dissolved in distilled water gives a clear solution which turns red litmus blue. Explain the phenomenon.
A person found that the cake prepared by him is hard and small in size. Which ingredient has he forgotten to add that would have caused the cake to rise and become light? Explain your answer.
A white chemical compound becomes hard on mixing with proper quantity of water. It is also used in surgery to maintain joints in a fixed position. Name the chemical compound.
When chlorine and sodium hydroxide being produced during the electrolysis of brine are allowed to mix, a new chemical is formed. Name the chemical and write its uses.
Write the name and formula of one salt each which contains:
two molecules of water of crystallisation
Write the name and formula of one salt each which contains:
five molecules of water of crystallisation
Write the name and formula of one salt each which contains:
ten molecules of water of crystallisation
How many molecules of water of crystallisation (per formula unit) are present in:
copper sulphate crystals?
How many molecules of water of crystallisation (per formula unit) are present in:
washing soda?
How many molecules of water of crystallisation (per formula unit) are present in:
gypsum?
Solutions for 2: Acids, Bases and Salts
![Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 chapter 2 - Acids, Bases and Salts Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 chapter 2 - Acids, Bases and Salts - Shaalaa.com](/images/9352831810-chemistry-science-english-class-10_6:7eeaf5db0f4a49c580d3c8746ff5ab36.jpg)
Lakhmir Singh solutions for Chemistry (Science) [English] Class 10 chapter 2 - Acids, Bases and Salts
Shaalaa.com has the CBSE Mathematics Chemistry (Science) [English] Class 10 CBSE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Lakhmir Singh solutions for Mathematics Chemistry (Science) [English] Class 10 CBSE 2 (Acids, Bases and Salts) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Lakhmir Singh textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry (Science) [English] Class 10 chapter 2 Acids, Bases and Salts are Similarities and Differences Between Acids and Bases, Preparation and Uses of Sodium Hydroxide, Preparation and Uses of Bleaching Powder, Preparation and Uses of Baking Soda, Preparation and Uses of Washing Soda, Preparation and Uses of Plaster of Paris, Acids, Bases (Alkalis), Indicators, Properties of Acids, Properties of Bases (Alkalis), Acid or a Base in a Water Solution, Strength of Acidic or Basic Solutions, Salts, Important Salts in Daily Life.
Using Lakhmir Singh Chemistry (Science) [English] Class 10 solutions Acids, Bases and Salts exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Lakhmir Singh Solutions are essential questions that can be asked in the final exam. Maximum CBSE Chemistry (Science) [English] Class 10 students prefer Lakhmir Singh Textbook Solutions to score more in exams.
Get the free view of Chapter 2, Acids, Bases and Salts Chemistry (Science) [English] Class 10 additional questions for Mathematics Chemistry (Science) [English] Class 10 CBSE, and you can use Shaalaa.com to keep it handy for your exam preparation.
