Advertisements
Advertisements
Question
Which gas is usually liberated when an acid reacts with a metal?
Solution
Hydrogen gas is usually liberated when an acid reacts with an active metal.
APPEARS IN
RELATED QUESTIONS
The soil in a field in highly acidic. Name two materials which can be added to this soil to reduce its acidity. Give the reason for your choice.
Wasp sting contains:
(a) a sugar solution
(b) an acidic liquid
(c) a salt solution
(d) an alkaline liquid
Do basic solutions also have H+(aq)? Why are they basic?
You are supplied with five solutions: A, B, C, D and E with pH values as follows: A = 1.8, B = 7, C = 8.5, D = 13, and E = 5
Classify these solutions as neutral, slightly or strongly acidic and slightly or strongly alkaline.
Which solution would be most likely to liberate hydrogen with magnesium powder. Give a word equation for each reaction.
State one observation for each of the following :
A small piece of zinc is added to dilute hydrochloric acid
Tartaric acid is a ______ acid.
If iron reacts with dilute sulphuric acid, what will be the products?
Match the chemical substances given in Column (A) with their appropriate application given in Column (B)
| Column (A) |
Column (B) |
| (A) Bleaching powder | (i) Preparation of glass |
| (B) Baking soda | (ii) Production of H2 and Cl2 |
| (C) Washing Soda | (iii) Decolourisation |
| (D) Sodium chloride | (iv) Antacid |
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
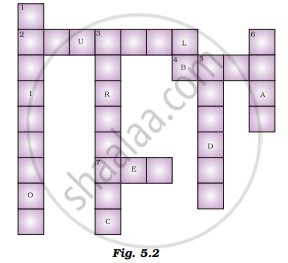
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Name the acid present in the given table.
