Advertisements
Advertisements
Question
What happens when a concentrated solution of sodium chloride (brine) is electrolysed? Write the equation of the reaction involved.
Solution
When electricity is passed through a concentrated solution of sodium chloride called brine, the solution decomposes to form sodium hydroxide, chlorine and hydrogen.
`2 NaCl(aq) + 2 H_2O (l)`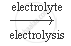 `2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
`2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
APPEARS IN
RELATED QUESTIONS
State what would you observe when Washing Soda Crystals are exposed to the atmosphere
From the list of the following salts choose the salt that most appropriately fits the
description is given in the following:
[AgCl, MgCl2, NaHSO4, PbCO3, ZnCO3, KNO3, Ca(NO3)2]
On heating this salt, a brown colored gas is evolved.
Why is the electrolysis of a concentrated solution of sodium chloride known as chlor-alkali process?
The aqueous solution of one of the following salts will turn red litmus to blue. This salt is:
(a) potassium sulphate
(b) sodium sulphate
(c) sodium chloride
(d) potassium carbonate
The pH values of three solutions A, B and C are given in the table. Answer the folloiwng questions:
| Solution | pH value |
| A | 12 |
| B | 2 |
| C | 7 |
Which solution will liberate CO2 when reacted with sodium carbonate?
Outline the steps required to convert hydrogen chloride to anhydrous iron (III) chloride. Write the equations for the reactions.
Answer the questions below relating your answers only to salts in the following list: Sodium chloride, anhydrous calcium chloride, copper sulphate-5-water?
- What name is given to the water in the compound copper sulphate-5-water?
- If copper sulphate-5-water is heated, anhydrous coppersulphate is formed. What is its colour?
- By what means, other than healing, could you dehydrate copper sulphate-5-water and obtain anhydrous coppersulphate?
- Which one of the salts in the given list is deliquescent?
Identify the following substance:
Gas C has an offensive smell like rotten eggs.
The chemical name of plaster of Paris is ______.
Salt which is formed by the partial replacement of hydrogen ions of an acid by a metal is called ______.
