Advertisements
Advertisements
प्रश्न
What happens when a concentrated solution of sodium chloride (brine) is electrolysed? Write the equation of the reaction involved.
उत्तर
When electricity is passed through a concentrated solution of sodium chloride called brine, the solution decomposes to form sodium hydroxide, chlorine and hydrogen.
`2 NaCl(aq) + 2 H_2O (l)`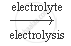 `2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
`2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
APPEARS IN
संबंधित प्रश्न
Name the raw material used for the production of caustic soda.
The aqueous solution of one of the following salts will turn red litmus to blue. This salt is:
(a) potassium sulphate
(b) sodium sulphate
(c) sodium chloride
(d) potassium carbonate
Which of the following salts will give an aqueous solution having pH of almost 7?
(a) NH4NO3
(b) NH4Cl
(c) CaCl2
(d) KCl
P and Q are aqueous solutions of sodium chloride and sodium hydroxide, respectively. Which of these will turn:
(a) blue litmus red?
A salt X when dissolved in distilled water gives a clear solution which turns red litmus blue. Explain the phenomenon.
Explain ‘salt hydrolysis’
name two salts which are basic.
Define the following term: Hygroscopy.
State what would you observe when :
Washing soda crystals are exposed to the atmosphere.
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Sodium zincate
