Advertisements
Advertisements
प्रश्न
What happens when a concentrated solution of sodium chloride (brine) is electrolysed? Write the equation of the reaction involved.
उत्तर
When electricity is passed through a concentrated solution of sodium chloride called brine, the solution decomposes to form sodium hydroxide, chlorine and hydrogen.
`2 NaCl(aq) + 2 H_2O (l)`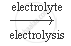 `2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
`2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
APPEARS IN
संबंधित प्रश्न
How would you show that blue copper sulphate crystals contain water of crystallisation?
Give one example of a salt which gives an aqueous solution having:
pH less than 7
Explain ‘salt hydrolysis’
Define the following term: Efflorescence.
Define the following term: Hygroscopy.
Name gases which has a sour taste.
Copy and complete the following table which refers to the action of heat on the carbonate:
| Carbonate | Colour of residue on cooling |
| Zinc Carbonate |
Differentiate hydrate and anhydrous salts with examples.
Salt is ______ in nature.
An aqueous solution of a salt turns blue litmus to red. The salt could be the one obtained by the reaction of ______.
