Advertisements
Advertisements
Question
Complete and balance the following chemical equations:
NaOH (aq) HCI (aq)→
Solution
NaOH(aq) + HCI(aq) → NaCI(aq) + H2O(l)
APPEARS IN
RELATED QUESTIONS
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
Write the chemical formula of soda ash?
Complete and balance the following chemical equations:
`Ca(OH)_2 + Cl_2 ->`
Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes.
Explain the following:
Lead carbonate does not react with dilute HCl.
Which of the following salts does not contain any water of crystallisation?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
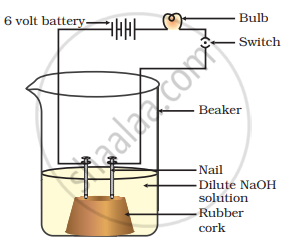
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
What happens when nitric acid is added to egg shell?
What property do acids and bases have in common? Explain it with an example.
