Advertisements
Advertisements
Question
What happens when bases react with non-metal oxides? Explain with the help of an example. What does this reaction tell us about the nature of non-metal oxides?
Solution
Bases react with non-metal oxides to form a salt and water.
Bases + Non-metal oxides Salt + Water
Example:
2NaOH(aq) + CO2 (g) → Na2 CO3 (aq) + H2O (l)
Here, sodium hydroxide (base) reacts with carbon dioxide (non-metal oxide) to give sodium carbonate (salt) and water.
This reaction shows that non-metal oxides are acidic in nature.
APPEARS IN
RELATED QUESTIONS
Name one natural source of the following acids:
Oxalic acid
How will you test for the presence of this gas?
Which chemical is injected into the skin of a person during an ant's sting?
How can the effect of these stings be neutralised?
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound is used as an antacid?
Name the acid present in vinegar.
Write balanced equation to satisfy the following statement:
\[\ce{Acid + Carbonate or bicarbonate -> Salt + water + Carbon dioxide}\]
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
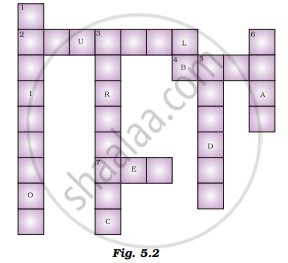
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Acids have a sour taste and they are soapy to touch.
Write the important uses of hydrochloric acid and sulphuric acids.
Complete the following equation.
Ca(OH)2 + ______ → ______ + H2O
