Advertisements
Advertisements
Question
When chlorine and sodium hydroxide being produced during the electrolysis of brine are allowed to mix, a new chemical is formed. Name the chemical and write its uses.
Solution
When chlorine and sodium hydroxide produced during the electrolysis of brine are allowed to mix, a new chemical called sodium hypochlorite (NaClO) is formed.
Sodium hypochlorite is a bleaching agent that is used in making household bleaches and for bleaching fabrics.
APPEARS IN
RELATED QUESTIONS
Write the chemical name of bleaching powder and write its properties.
Name one compound of calcium which is used for removing the colour of a coloured cloth.
Name the substance obtained by the action of chlorine on solid (dry) slaked lime.
What is "baking powder"? How does it make the cake soft and spongy?
What is the common name of the compound CaOCl2?
How is bleaching powder prepared? Write chemical equation of the reaction involved in the preparation of bleaching powder.
What happens when bleaching powder reacts with dilute sulphuric acid? Give equation of the reaction involved.
Give a scientific explanation.
Bleaching powder has the odour of chlorine.
The reaction between MnO2 with HCl is depicted in the following diagram. It was observed that gas with bleaching abilities was released.
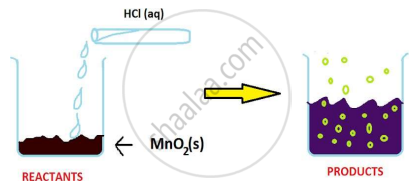
The chemical reaction between MnO2 and HCl is an example of:
Assertion (A): Reaction of Quicklime with water is an exothermic reaction.
Reason (R): Quicklime reacts vigorously with water releasing a large amount of heat.
