Advertisements
Advertisements
Question
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Solution
Methyl orange: Methyl orange changes to red when added to an acid.
APPEARS IN
RELATED QUESTIONS
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Compounds such as alcohols and glucose also contain hydrogen but are not categorized as acids. Describe an activity to prove it.
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
red cabbage extract
What happens when carbon dioxide gas is passed through lime water for a short time?
Write equations of the reactions involved.
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
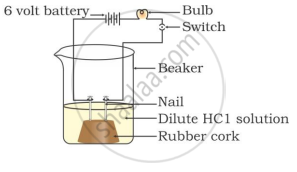
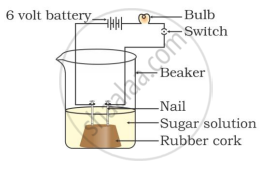
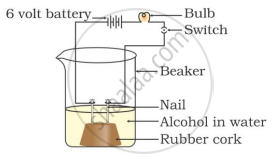
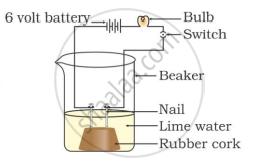
In which of the following setups would the bulb glow?
Identify the correct representation of reaction occurring during chloralkali process
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, It gives a compound Z, used for disinfecting drinking water. Identity X, Y, G and Z.
- A compound 'A' with a molecular formula of \[\ce{C2H4O2}\] reacts with a base to give salt and water. Identify 'A', state its nature and the name of the functional group it possesses. Write chemical equation for the reaction involved.
- When the above stated compound 'A' reacts with another compound 'B' having molecular formula \[\ce{C2H6O}\] in the presence of an acid, a sweet smelling compound is 'C' formed.
- Identify 'B' and 'C'.
- State the role of acid in this reaction.
- Write chemical equation for the reaction involved.