Advertisements
Advertisements
Question
- A compound 'A' with a molecular formula of \[\ce{C2H4O2}\] reacts with a base to give salt and water. Identify 'A', state its nature and the name of the functional group it possesses. Write chemical equation for the reaction involved.
- When the above stated compound 'A' reacts with another compound 'B' having molecular formula \[\ce{C2H6O}\] in the presence of an acid, a sweet smelling compound is 'C' formed.
- Identify 'B' and 'C'.
- State the role of acid in this reaction.
- Write chemical equation for the reaction involved.
Solution
- Since acid and base react to form salt and water, compound A will naturally be acidic due to the presence of a carboxylic acid group.
Chemical equation for acid and base reaction:
\[\ce{Acid + Base-> Salt + Water}\]
\[\ce{H X + M OH -> MX + HOH}\]
\[\ce{CH3COOH(aq) + NaOH(aq) -> CH3COONa(aq) + H2O(l)}\]
Thus, Compound A is \[\ce{CH3COOH}\] it is acidic in nature containing a carboxyl functional group. - In a process known as Fischer esterification, carboxylic acids combine with alcohols to create esters. An acid catalyst is required, and alcohol serves as a reaction solvent.
- Compound B is Ethanol and C is Ethyl acetate.
- HCl act as catalyst.
- A sweet-smelling compound formed by reacting acetic acid with ethanol in the presence of hydrochloric acid is ethyl acetate.
\[\begin{array}{cc} \ce{CH3 - C - OH + HOC2H5 ->[H+] CH3 - C - OC2H5}\\ ||\phantom{..............................}||\phantom{..}\\ \phantom{...}\ce{O}\phantom{..........................}\ce{\underset{Ethyl acetate}{O}} \end{array}\]
APPEARS IN
RELATED QUESTIONS
Why do acids not show acidic behaviour in the absence of water?
Lime water turns milky when __________ gas is passed through it.
(a) H2
(b) CO
(c) CO2
(d) SO2
What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced chemical equation of the reaction involved.
Which gas is liberated when dilute hydrochloric acid reacts with sodium carbonate?
Explain with an example. Give the chemical equation of the reaction which takes place.
You have been provided with three test-bubes. One of these test-tubes contains distilled water and the other two contain an acidic and a basic solution respectively. If you are given only blue litmus paper, how will you identify the contents of each test-tube?
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
Answer the following question.
Blue litmus solution is added to two test tubes A and B containing dilute HCl and NaOH solution respectively. In which test tube a colour change will be observed? State the colour change and give its reason.
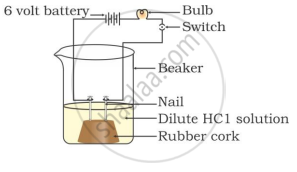
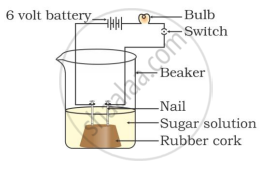
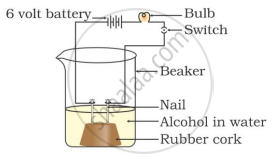
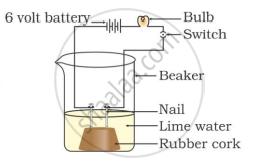
In which of the following setups would the bulb glow?
Which of the following is(are) true when HCl (g) is passed through water?
- It does not ionise in the solution as it is a covalent compound.
- It ionises in the solution
- It gives both hydrogen and hydroxyl ions in the solution
- It forms hydronium ion in the solution due to the combination of hydrogen ion with water molecule