Advertisements
Advertisements
Question
Name the compound formed when ethanol is heated at 443 K in the presence of conc.
State the role of conc.
Solution
Ethene is created when 443K ethanol is heated with excess concentrated sulphuric acid.
The mechanism of acid dehydration of ethanol to yield ethene is:
-
- The first step is to protonate the oxygen atom of the -OH group.
- The second step is the loss of a water molecule to generate carbonium ions.
- Deprotonation, the final step, creates a carbon-carbon double bond.
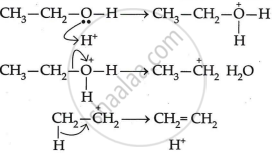
Sulphuric acid in concentrated form has a dehydrating effect. In light of this, ethanol is produced at 433 K with extra cone. To create ethene,
APPEARS IN
RELATED QUESTIONS
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
Draw the structures for the following compounds:
Propanal
Draw the structures for the following compounds:
Butanal
How would you convert of thanol into ethene?
How is acetic acid prepared from ethanol?
Give two uses each of:
(i) Methane (ii) ethane
Write the equations of chlorination of ethane
Name a solid which can be used, instead of conc H2SO4 to prepare ethylene by dehydration of ethanol.
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above list, name the compound with
Give the balance chemical equation of the following reaction:
Combustion of ethanol.
