Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
उत्तर
Ethene is created when 443K ethanol is heated with excess concentrated sulphuric acid.
The mechanism of acid dehydration of ethanol to yield ethene is:
-
- The first step is to protonate the oxygen atom of the -OH group.
- The second step is the loss of a water molecule to generate carbonium ions.
- Deprotonation, the final step, creates a carbon-carbon double bond.
\[\ce{\underset{Ethanol}{2CH3CH2OH}->[\Delta][Conc.sulphuric acid] \underset{Ethene}{2CH2 = CH2} + \underset{Water}{2H2O}}\]
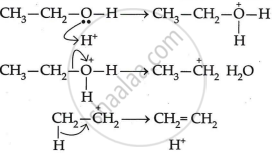
Sulphuric acid in concentrated form has a dehydrating effect. In light of this, ethanol is produced at 433 K with extra cone. To create ethene, \[\ce{H2SO4}\] undergoes a dehydration process.
APPEARS IN
संबंधित प्रश्न
What is the molecular formula and structure of the alcohol which can be thought to be derived from pentane?
State two used of ethanol (other than as a fuel).
How is the absolute alcohol obtained?
Give three uses of ethanol
Addition reactions and substitution reactions are types of organic reactions.Which type of reaction is shown by?
Ethene?
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above, name the homologue of the homologous series with the general formula CnH2n.
Complete the following activity.
| Boiling point of ethanol | → | |
| General name of ethanol | → | |
| Use of ethanol | → | |
| Boiling point of ethanoic acid | → | |
| Melting point of pure ethanoic acid | → |
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Write the chemical equation for its reaction with ethanol to form compound ‘B’.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
