Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
उत्तर
Ethene is created when 443K ethanol is heated with excess concentrated sulphuric acid.
The mechanism of acid dehydration of ethanol to yield ethene is:
-
- The first step is to protonate the oxygen atom of the -OH group.
- The second step is the loss of a water molecule to generate carbonium ions.
- Deprotonation, the final step, creates a carbon-carbon double bond.
\[\ce{\underset{Ethanol}{2CH3CH2OH}->[\Delta][Conc.sulphuric acid] \underset{Ethene}{2CH2 = CH2} + \underset{Water}{2H2O}}\]
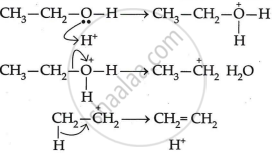
Sulphuric acid in concentrated form has a dehydrating effect. In light of this, ethanol is produced at 433 K with extra cone. To create ethene, \[\ce{H2SO4}\] undergoes a dehydration process.
APPEARS IN
संबंधित प्रश्न
What is meant by a functional group? Explain with an example.
Write the chemical equation of the reaction which takes place during the burning of ethanol in air.
Explain why, methanol is much more dangerous to drink than ethanol.
Name the gas evolved when ethanol reacts with sodium.
Which of the following can damage optic nerve leading to blindness, if taken internally?
(a) CH3COOH
(b) C2H5OH
(c) NaHCO3
(d) CH3OH
How is the absolute alcohol obtained?
How will you bring about following conversions:
(a) Methane to methanol
(b) Ethane to ethanol (acetaldehyde)
(c) Methane to methanoic acid
Convert ethanol into ethene using hot conc. H2SO4. Give only balanced equation.
Ethanol is soluble in water in all proportions.
Draw the electron dot structure of ethyne and also draw its structural formula
