Advertisements
Advertisements
Question
Why do HCl, HNO3, etc., show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Solution 1
- When HCl or HNO3 are mixed with water then they dissolve in water to form H+ or H3O+ ions which shows their acidic character.
- For example just see the following reactions:
- When alcohols and glucose are mixed with water then they do not dissolve to form ions. Hence they do not show acidic character.
Solution 2
- Acids are substances that can dissociate on the dissolving of water, which results in production of hydrogen ions. Some acids show acidic character as they dissociate in the aqueous solution which results in the production of hydrogen ions (acids like HCl, HNO3).
- Compounds similar to glucose or alcohol also do contain hydrogen element but they do not show signs of acidic nature. This is due to the fact that the hydrogen in them will not separate as like the hydrogen in the acids. They will not separate to become hydrogen ions, on dissolving in the water.
APPEARS IN
RELATED QUESTIONS
Dilute NaOH can be tested with
(a) red litmus paper
(b) blue litmus paper
(c) lime water
(d) Na2CO3
From which plant is litmus paper or litmus solution obtained?
- Moss
- Rose
- Hibiscus
- Lichen
How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with iron filings.
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
Fill in the blank in the following sentences:
Substances do not show their acidic properties without.......................... .
A solution reacts with zinc granules to give a gas which burns with a 'pop' sound. The solution contains:
(a) Mg(OH)2
(b) Na2CO3
(c) NaCl
(d) HCl
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
What ions are present in the solutions of following substances? (write the symbols only)
Magnesium hydroxide
Define an acid and a base. Give two examples of each.
State two uses each of the following compounds:
Sodium hydroxide
Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes.
When _______________ is passed through fresh lime water, it turns milky.
A sample of soil is mixed with water and allowed to settle. The clear supernatant solution turns the pH paper yellowish-orange. Which of the following would change the colour of this pH paper to greenish-blue?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
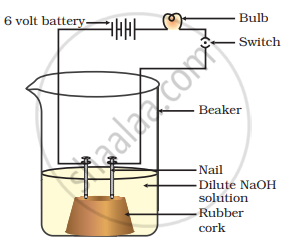
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Which of the following statements is not correct?
Which of the following are present in a dilute aqueous solution of hydrochloric acid?
Vinegar is ______ in taste.
What property do acids and bases have in common? Explain it with an example.
The metal that will not produce hydrogen gas when reacted with dilute acids.
