Advertisements
Advertisements
Question
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made?

- In place of zinc granules, same amount of zinc dust is taken in the test tube
- Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
- In place of zinc, copper turnings are taken
- Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
Solution
- Hydrogen gas would evolve at a greater speed because zinc dust provides more surface area as compared to zinc granules.
- Both would react in the same way. There would be no effect on the volume of the gas evolved.
- Copper does not react with either dilute HCl or dilute H2SO4. No gas would evolve in both the cases.
- Hydrogen gas would evolve in this case also.
`"Zn" + 2"NaOH" overset("heat")(->) underset("Sod. zincate")("Na"_2"ZnO"_2) + "H"_2`
APPEARS IN
RELATED QUESTIONS
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
What ions are present in the solutions of following substances? (write the symbols only)
Nitric acid
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
Write the chemical formula of soda ash?
With which substance should chlorine be treated to get bleaching powder?
Which of the following substance will not give carbon dioxide on treatment with dilute acid?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
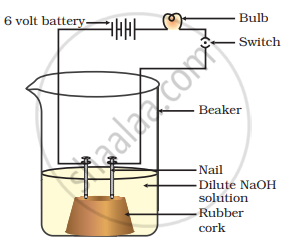
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Which of the following statements is true for acids?
Can we taste acids and bases to identify them?
