Advertisements
Advertisements
प्रश्न
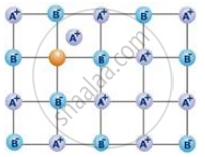
Examine the given defective crystal:

Answer the following questions :
(i) What type of stoichiometric defect is shown by the crystal?
(ii) How is the density of the crystal affected by this defect?
(iii) What type of ionic substances show such defect?
उत्तर
(i) Schottky defect is shown by the mentioned crystal, as equal number of cations and anions are missing in the crystal lattice.
(ii) This defect leads to decrease in density, as equal number of the cations and anions are missing from the crystal lattice.
(iii) This kind of defect is shown by those ionic substance in which the cations and anions are of almost similar sizes.
Examples: NaCl, KCl and CsCl.
APPEARS IN
संबंधित प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Ionic solids containing large differences in sizes of ions show ____________.
Point defect is also known as ____________.
Frenkel defects are not found in alkali metal halides because ____________.
The radius of Cs+ is 169 pm and Cl− is 181 pm. The radius ratio is ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Which of the following defects is also known as dislocation defect?
Schottky defect is noticed in ______
