Advertisements
Advertisements
प्रश्न
Explain in brief the experimental proof which led to the discovery of –
Atomic nucleus
उत्तर
Discovery of Atomic Nucleus: Lord Rutherford in 1911 directed alpha particles (42He) towards gold foil. The deflection of the alpha particles was observed and concluded that there was a nucleus at the centre of atom which was positively charged dense very small space (solid) called the nucleus.
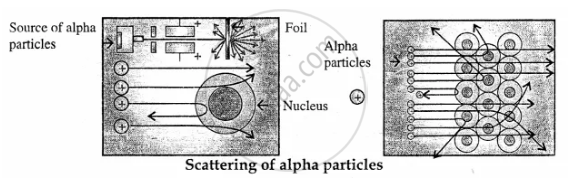
APPEARS IN
संबंधित प्रश्न
Give the following a suitable word/phrase.
The number of protons present in the nucleus of an atom.
Multiple Choice Questions
The number of electrons in an atom is equal to number of
Draw the diagrams representing the atomic structures of the following: Nitrogen.
Tick the most appropriate answer
Which one of the following scientists put forward the theory regarding the extra-nuclear structure of the atom?
- James Chadwick
- John Dalton
- Rutherford
- Niels Bohr
Tick the most appropriate answer.
Which of the following is used as a coolant in nuclear reactor?
- liquid sodium
- liquid nitrogen
- cadmium steel rods
- plutonium
ANSWER IN DETAIL
What are the various components of a nuclear power plant?
ANSWER IN DETAIL
How are radioactive radiations useful in medicines ?
Explain in brief – Lod Rutherford’s experiment which led to the discovery of the atomic nucleus.
‘Electrons revolve around the nucleus in fixed orbits or shells called energy levels’. State how these energy levels are represented.
What is meant by the term nucleus
