Advertisements
Advertisements
प्रश्न
Explain in brief the experimental proof which led to the discovery of –
Atomic nucleus
उत्तर
Discovery of Atomic Nucleus: Lord Rutherford in 1911 directed alpha particles (42He) towards gold foil. The deflection of the alpha particles was observed and concluded that there was a nucleus at the centre of atom which was positively charged dense very small space (solid) called the nucleus.
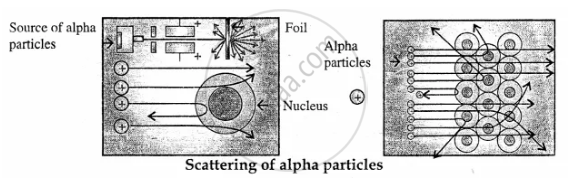
APPEARS IN
संबंधित प्रश्न
Fill in the blank
The ..........................of an atom is very hard and dense.
Give the following a suitable word/phrase.
The number of protons present in the nucleus of an atom.
How does the existence of isotopes contradict Dalton’s atomic theory?
Complete the table below by identifying A, B, C, D, E and F.
FILL IN THE BLANK
The protons and neutrons are held firmly in the nucleus by strong .........................
FILL IN THE BLANK
Nuclear energy can be used to make ..........................
ANSWER IN DETAIL
What are the various components of a nuclear power plant?
State Dalton's atomic theory.
How was it shown that atom has empty space?
Explain in brief – Lod Rutherford’s experiment which led to the discovery of the atomic nucleus.
