Advertisements
Advertisements
प्रश्न
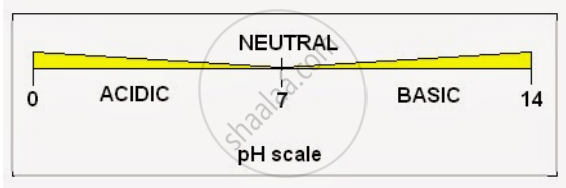
Explain the pH scale with the proper diagram.
उत्तर
- pH helps in measuring hydrogen ion concentration in solutions. In pH, p stands for “potenz” means “strength” in German.
- The scale reads from O (zero) (most acidic) to 14 (most basic).
- The value of pH indicates the acidic or basic nature of a solution. The strength of the base is represented by pOH.
- When the pH value is between 0 to 7, the solution is acidic in nature. At value 7, the solution is neutral and between 7 to 14 the nature of the solution becomes alkaline/ basic.
- Lower the pH of an acid, greater is the concentration of H+ ions, greater the pH of a base, greater is the concentration of OH ions.
APPEARS IN
संबंधित प्रश्न
The pH of rainwater is 7.
Which one of the following types of medicines is used for treating indigestion?
Fill in the blank
Magnesia is used in making __________.
A solution 'X' gives orange colour when a drop of it falls on pH paper, while another solution 'Y' gives bluish colour when a drop of it falls on pH paper. What is the nature of both solutions? Determine the pH of solutions 'X' and 'Y'.
Give the tests to identify Acids and Bases.
Tomato is a natural source of which acid?
Which of the following statements is correct about an aqueous solution of an acid and of a base?
(i) Higher the pH, stronger the acid
(ii) Higher the pH, weaker the acid
(iii) Lower the pH, stronger the base
(iv) Lower the pH, weaker the base
What is the pH range of our body?
Rain is called acid rain when it:
An aqueous solution with pH = 1 is ___________.
Which of the following is used for the dissolution of gold?
Ag2S reacts with H2SO4 to form ____________.
The graph given below depicts a neutralization reaction (acid + alkali → salt + water). The pH of a solution changes as we add an excess acid to an alkali.

Which letter denotes the area of the graph where both acid and salt are present?
Assertion: Fresh milk in which baking soda is added, takes a longer time to set as curd.
Reason: Baking soda decreases the pH value of fresh milk to below 6.
Frothing in Yamuna:
The primary reason behind the formation of the toxic foam is the high phosphate content in the wastewater because of detergents used in dyeing industries, dhobi ghats, and households. Yamuna's pollution level is so bad that parts of it have been labelled 'dead' as there is no oxygen in it for aquatic life to survive.

Predict the pH value of the water of river Yamuna if the reason for froth is the high content of detergents dissolved in it.
Which of the following statements is correct about an aqueous solution of an acid and of a base?
- Higher the pH, stronger the acid
- Higher the pH, weaker the acid
- Lower the pH, stronger the base
- Lower the pH, weaker the base
Equal volumes of hydrochloric acid and sodium hydroxide solutions of same concentration are mixed and the pH of the resulting solution is checked with a pH paper. What would be the colour obtained?

The pH of stomach fluid is ______.
pH stands for ______ in a solution.
Correct the wrong statement given below:
An acid is the compound which are capable of forming hydroxyl ions (OH– ) in aqueous solution.
Write notes on the importance of pH in everyday life.
Acid present in tomato is ______.
How does Paramecium obtain is food?
List the role of each of the following in our digestive system:
- Hydrochloric acid
- Trypsin
- Muscular walls of stomach
- Salivary amylase
Mohan has three solutions P, Q and R having a pH of 13, 5 and 2 respectively. Which of the above solutions P, Q or R will contain molecules as well as ions?
Rohit has solution X, Y and Z that has pH 2, 7 and 13 respectively.
Which solution will liberate ammonia gas when reacted with ammonium chloride?
