Advertisements
Advertisements
प्रश्न
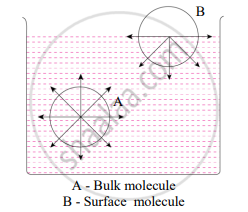
Explain the Adsorption is a surface phenomenon.
उत्तर
Consider the surface of a liquid or a solid.
- The molecular forces at the surface of a liquid are unbalanced or in an unsaturation state.
- In solids, the ions or molecules at the surface of a crystal do not have their forces satisfied by close contact with other particles.
- Because of the unsaturation, solid and liquid surfaces tend to attract gases or dissolved substances with which they come in close contact. Thus, the substance accumulates on the surface of a solid or liquid i.e., the substance gets adsorbed on the surface. Hence, adsorption is a surface phenomenon.
APPEARS IN
संबंधित प्रश्न
Define adsorption.
Why students can read blackboard written by chalks?
Aqueous solution of raw sugar, when passed over beds of animal charcoal, becomes colourless. Explain.
A graph is plotted between log (x/m) and log P according to the equation `x/"m" - "kP"^(1/"n")`
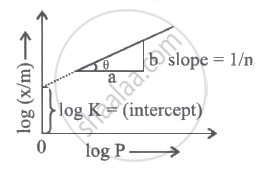
Which of the following statements about this graph is not correct?
Which one of the following is correct for the adsorption of a gas at a given temperature on a solid surface?
For adsorption of gas over solid surface following data is obtained at 300 K.
| Pressure of gas (mm of Hg) |
100 | 25 |
| Amount adsorged per kg of charcoal |
3 gm | 1.5 gm |
The slope of the graph between log P vs log x/m will be ______.
[x/m and P are in same units as given in question.]
The process by which sites of adsorbent are made free so, that more adsorbent can occupy them is called ______.
