Advertisements
Advertisements
प्रश्न
Fill in the blank:
On addition of silver nitrate to hydrochloric acid ___________ precipitate is formed which is soluble in ____________
उत्तर
On addition of silver nitrate to hydrochloric acid silver chloride precipitate is formed which is soluble in HCl.
APPEARS IN
संबंधित प्रश्न
State your observation when dilute hydrochloric acid is added to a lead nitrate solution and the mixture is heated.
Explain why anhydrous \[\ce{HCl}\] is a poor conductor while aqueous \[\ce{HCl}\] is an excellent conductor.
Write an equation for the reaction of hydrochloric acid on copper oxide.
Name the following:
A metallic oxide which reacts with hydrochloric acid to give a coloured solution.
State the use of aqua-regia.
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Complete the table by writing the gas evolved in each case and its odour.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Magnesium sulphite
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
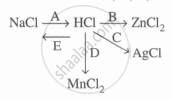
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
