Advertisements
Advertisements
प्रश्न
From the following elements :
4Be; 9F; 19K; 20Ca
(i) Select the element having one electron is the outermost shell.
(ii) two elements of the same group.
Write the formula of and mention the nature of the compound formed by the union of 19K and element X(2, 8, 7).
उत्तर
(i) Among the given elements, 19K has one electron in the outermost shell. Its electronic configuration is 2, 8, 8, 1.
(ii) The elements 4Be and 20Ca belong to the same group, that is, Group 2. They both have two electrons each in their outermost shells.
Electronic configuration of 4Be is 2, 2.
Electronic configuration of 20Ca is 2, 8, 8, 2.
Atomic number of element X = 17
Electronic configuration of element X is 2, 8, 7.
Number of valence electrons in X = 7
Valency of X = 8 − 7 = 1
Atomic number of K = 19
Electronic configuration of K is 2, 8, 8, 1.
Number of valence electrons in K = 1
Valency of K = 1
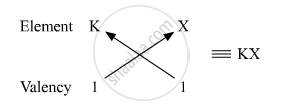
Therefore, the formula of the compound formed when K and element X combine is KX. The compound KX is of ionic nature.
APPEARS IN
संबंधित प्रश्न
The atomic masses of three elements A, B and C having similar chemical properties are 7, 23 and 39, respectively.
- Calculate the average atomic mass of elements A and C.
- Compare the average atomic mass with atomic mass of B.
- What could the elements A, B and C be?
The elements Be, Mg and Ca each having two electrons in their outermost shells are in periods 2, 3, and 4 respectively of the modern periodic table. Answer the following questions, giving justification in each case:-
(i) Write the group to which these elements belong.
(ii) Name the least reactive element.
(iii) Name the element having largest atomic radius.
An element Z has atomic number 16. Answer the following questions on Z:
1) State the period and group to which Z belongs
2) Is Z a metal or a non-metal?
3) State the formula between Z and hydrogen
4) What kind of a compound is this?
name the elements in period 1.
First ionization enthalpy of two elements X and Y are 500 KJ mol-1 and 375 KJ mol-1 respectively. Comment about their relative position in a group as well as in a period.
What is meant by periodicity of elements?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The similarities in the properties of elements belonging to a group are because they have the same ______
Atomic number of an element is 16. State
- the period to which it belongs
- the number of valence electrons
- whether it is a metal or non-metal
Choose the most appropriate answer from the following list of oxides which fit the description.
An oxide which dissolves in water forming an acid.
Based on the group valency of element write the molecular formula of the following compound giving justification:
Compound formed when an element A of group 2 combines with an element B of group seventeen.
