Advertisements
Advertisements
प्रश्न
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Electrical energy
उत्तर
Electrical Energy:
It dissociates the reactant in molten or solution state, into ions, which are charged particles and hence respective products are obtained.
Example:
Electrolysis of acidified water.
\[\ce{2H2O ->[electric][current] 2H2 [g] + O2[g]}\]
APPEARS IN
संबंधित प्रश्न
Balance the given chemical equation:
NH3 → N2 + H2
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in temperature.
Give chemical equation for the reaction involved in the above case.
Balance the following chemical equation:
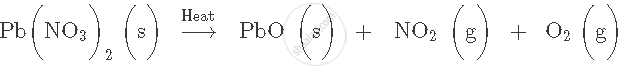
Balance the following equation. Also name the product formed.
PbCO3 → PbO +CO2
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Is the equation given above a balanced equation. Give reasons.
Give an example of a chemical equation in which two reactants form:
one product
Balance the following simple equation:
H2S + Cl2 → S + HCl
Balance the following simple equation:
K + CO2 → K2O + C
Write a balanced equation for the following word equation:
Nitrogen dioxide + Water + Oxygen → Nitric acid
What is a product in a chemical equation?
