Advertisements
Advertisements
प्रश्न
Give two reactions showing oxidising property of concentrated H2SO4. Write the structures of HClO3 and HClO4.
थोडक्यात उत्तर
उत्तर
Metals and non-metals both are oxidised by hot, concentrated sulphuric acid which itself gets reduced to SO2.
- \[\ce{\underset{Copper}{Cu} + \underset{\underset{Acid(Conc.)}{Sulphuric}}{2H2SO4} -> \underset{\underset{Sulphate}{Copper}}{CuSO4} + 2H2O}\]
- \[\ce{\underset{Sulphuric}{S} + \underset{Sulpuric acid(Conc.)}{2H2SO4} -> \underset{Sulphur dioxide}{3SO2} + 2H2O}\]
Structures of HClO3 and HClO4
Chloric acid. HClO3
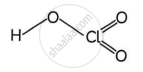
Perchloric acid HClO4
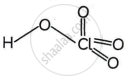
shaalaa.com
Chemical Properties of Elements of Groups 16, 17 and 18
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
