Advertisements
Advertisements
Question
Give two reactions showing oxidising property of concentrated H2SO4. Write the structures of HClO3 and HClO4.
Answer in Brief
Solution
Metals and non-metals both are oxidised by hot, concentrated sulphuric acid which itself gets reduced to SO2.
- \[\ce{\underset{Copper}{Cu} + \underset{\underset{Acid(Conc.)}{Sulphuric}}{2H2SO4} -> \underset{\underset{Sulphate}{Copper}}{CuSO4} + 2H2O}\]
- \[\ce{\underset{Sulphuric}{S} + \underset{Sulpuric acid(Conc.)}{2H2SO4} -> \underset{Sulphur dioxide}{3SO2} + 2H2O}\]
Structures of HClO3 and HClO4
Chloric acid. HClO3
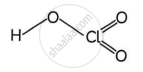
Perchloric acid HClO4
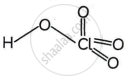
shaalaa.com
Chemical Properties of Elements of Groups 16, 17 and 18
Is there an error in this question or solution?
