Advertisements
Advertisements
प्रश्न
How is carbolic acid prepared from chlorobenzene?
उत्तर
1. Dow’s process:
i. When chlorobenzene is heated with excess of aqueous sodium hydroxide (NaOH) at 613 K under a pressure of 300 atmosphere, sodium phenoxide is formed.
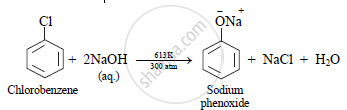
ii. Sodium phenoxide when hydrolysed using dilute hydrochloric acid (HCl), phenol is obtained.
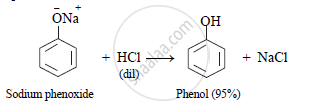
OR
Phenol can also be obtained by passing CO2 (carbon dioxide) gas, through aqueous solution of sodium phenoxide.
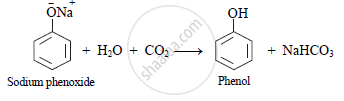
2. Raschig process (Industrial method):
i. When chlorobenzene is treated with steam (H2O) at 698 K in the presence of Ca3(PO4)2 or SiO2 as a catalyst, phenol is obtained.

ii. The hydrolysis can also be carried out by using water in the presence of copper catalyst at 673 K and under pressure.
Notes
(Any one method)
APPEARS IN
संबंधित प्रश्न
Write the structures of A and B in the following reactions
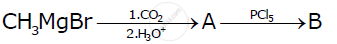
Show how the following compound can be converted to benzoic acid.
Bromobenzene
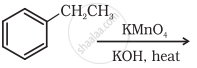
Show how the following compound can be converted to benzoic acid.
Phenylethene (Styrene)
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagent.
Tollens’ reagent
An organic compound (A) (molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives but-1-ene. Write equations for the reactions involved.
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
m-Nitrobenzoic acid
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Phenylacetic acid
How will you bring about the following conversion in not more than two steps?
Benazaldehyde to α-Hydroxyphenylacetic acid
Complete the synthesis by giving missing starting material, reagent or product.
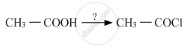
Name the reagents used in the following reactions:
The reagent which does not react with both, acetone and benzaldehyde.
Which is the most suitable reagent for the following conversion?
\[\begin{array}{cc}
\phantom{....................}\ce{O}\phantom{.....................................}\ce{O}\phantom{.}\\
\phantom{....................}||\phantom{......................................}||\phantom{.}\\
\phantom{}\ce{CH3 - CH = CH - CH2 - C - CH3 -> CH3 - CH = CH - CH2 - C - OH}\phantom{.}
\end{array}\]
Through which of the following reactions number of carbon atoms can be increased in the chain?
(i) Grignard reaction
(ii) Cannizaro’s reaction
(iii) Aldol condensation
(iv) HVZ reaction
Assertion: Aldehydes and ketones, both react with Tollen’s reagent to form silver mirror.
Reason: Both, aldehydes and ketones contain a carbonyl group.
Which of the following substance produced acetaldehyde on dry distillation?
Match List - I with List - II.
| List - I | List - II | ||
| (a) |  \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] \[\ce{->[CO,HCl][Anhyd. AlCl3/CuCl]}\] |
(i) | Hell-Volhard-Zelinsky reaction |
| (b) |
\[\begin{array}{cc}
\ce{O}\phantom{.................}\\ ||\phantom{.................}\\ \ce{R - C - CH3 + NaOX ->} \end{array}\] |
(ii) | Gattermann-Koch reaction |
| (c) | \[\ce{R - CH2 - OH + R'COOH ->[Conc. H2SO4]}\] | (iii) | Haloform reaction |
| (d) | \[\ce{R - CH2COOH ->[(i) X2/Red P][(ii) H2O]}\] | (iv) | Esterification |
Choose the correct answer from the options given below.
The end product Y in the sequence of reaction:
\[\ce{RX ->[CN^-] X ->[NaOH] Y}\] is:
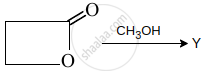
Y is:
A compound 'X' with molecular formula C3H8O can be oxidised to a compound 'Y' with the molecular formula C3H6O2 'X' is most likely to be ______.
