Advertisements
Advertisements
प्रश्न
How do you explain the absence of the aldehyde group in the pentaacetate of D-glucose?
उत्तर
The cyclic hemiacetal form of glucose contains an OH group at C-l which gets hydrolysed in aqueous solution to produce an open-chain aldehydic form, which then reacts with NH2OH to form the corresponding oxime. Thus, glucose contains an aldehydic group. However, when glucose is reacted with acetic anhydride, the OH group at C-l along with the other OH groups at C-2, C-3, C-4 and C-6 forms a pentaacetate. Since the pentaacetate of glucose does not contain a free OH group at C-l, it cannot get hydrolysed in aqueous solution to produce an open-chain aldehydic form and hence glucose pentaacetate does not react with NH2OH to form glucose oxime. Hence, glucose pentaacetate does not contain the aldehdye group.

APPEARS IN
संबंधित प्रश्न
What are monosaccharides?
Draw ring structure of α - D - (+) - glucopyranose.
Write the name of two monosaccharides obtained on hydrolysis of lactose sugar.
Write the product obtained when D-glucose reacts with H2N − OH.
Which one of the following is a monosaccharide:
starch, maltose, fructose, cellulose
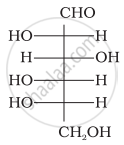
The letters ‘D’ or ‘L’ before the name of a stereoisomer of a compound indicate the correlation of configuration of that particular stereoisomer. This refers to their relation with one of the isomers of glyceraldehyde. Predict whether the following compound has ‘D’ or ‘L’ configuration.
Assertion: All naturally occurring α-aminoacids except glycine are optically active.
Reason: Most naturally occurring amino acids have L-configuration.
Naturally occurring glucose is called as ______.
