Advertisements
Advertisements
प्रश्न
How does the branching of hydrocarbon chain of synthetic detergents affect their biodegradability?
उत्तर
Straight chain hydrocarbons in synthetic detergent leads to easy biodegradability because the branched-chain hydrocarbon tail is a source of pollution. Therefore, lesser the branching more is the biodegradability.
APPEARS IN
संबंधित प्रश्न
Explain the following terms with suitable examples - Anionic detergents
Label the hydrophilic and hydrophobic parts in the following compounds.
CH3(CH2)10CH2OSO3 –Na+
What type of detergent are used in toothpastes?
Polyethyleneglycols are used in the preparation of which type of detergents?
Which of the following are anionic detergents?
(i) Sodium salts of sulphonated long-chain alcohol.
(ii) Ester of stearic acid and polyethylene glycol.
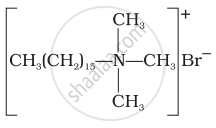
(iii) Quarternary ammonium salt of amine with acetate ion.
(iv) Sodium salts of sulphonated long-chain hydrocarbons.
Which of the following statements are correct?
(i) Cationic detergents have germicidal properties.
(ii) Bacteria can degrade the detergents containing highly branched chains.
(iii) Some synthetic detergents can give foam even in ice cold water.
(iv) Synthetic detergents are not soaps.
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
(iv) |
(d) Soap |
Explain the following term with suitable examples.
cationic detergents
Explain the following terms with suitable examples Cationic detergents