Advertisements
Advertisements
प्रश्न
Ice is more effective in cooling than the ice-water. Explain.
उत्तर
Each 1 g of ice absorbs nearly 336 J of heat when melts to water at 0°C. Hence, it is more effective in cooling than the ice-water.
APPEARS IN
संबंधित प्रश्न
Name the law on which the principle of mixtures is based
1300 J of heat energy is supplied to raise the temperature of 6.5 kg of lead from 20° C to 40°C. Calculate the specific heat capacity of lead.
0.5 kg of lemon squash at 30° C is placed in a refrigerator which can remove heat at an average rate of 30 J s−1. How long will it take to cool the lemon squash to 5°C? Specific heat capacity of squash = 4200 J g−1K−1.
200 g mass of certain metal of 83°C is immersed in 300 g of water at 30°C the final temperature
is 33°C. Calculate the specific heat capacity of the metal Assume that the specific heat capacity
of water is `4.2 J g^-1K^-1`
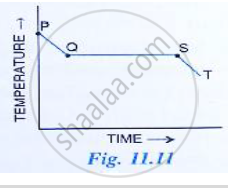
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
Find the result of mixing 10 g of ice at - 10℃ with 10 g of water at 10℃. Specific heat capacity of ice = 2.1 J kg-1 K-1, Specific latent heat of ice = 336 J g-1 and specific heat capacity of water = 4.2 J kg-1 K-1.
(i) Name the physical quantity which is measured in calories.
(ii) How is calorie related to the S.I. unit of that quantity ?
Why does atmospheric temperature fall after hail storm?
40g of ice at 0°C is used to bring down the temperature of a certain mass of water at 60°C to 10°C. Find the mass of water used.
[Specific heat capacity of water = 4200 J kg-1 °C-1]
[Specific latent heat of fusion of ice = 336 × 103 J kg-1]
The process of converting a substance from solid state to gaseous state is called condensation.
