Advertisements
Advertisements
प्रश्न
उत्तर
\[\ce{\underset{(Ethyl alcohol)}{CH3CH2OH} ->[red 'P'][Br2] \underset{(A)}{\underset{(Ethyl bromide)}{CH3 - CH2 - Br}}}\]
\[\ce{\underset{(A)}{\underset{(Ethyl bromide)}{CH3 - CH2 - Br}} ->[KCN][alc.] \underset{(B)}{\underset{(Ethyl cyanide)}{CH3 - CH2 - CN}}}\]
\[\ce{\underset{(B)}{\underset{(Ethyl cyanide)}{CH3 - CH2 - CN}} ->[LiAH4][Ether] \underset{(C)}{\underset{(Propan-1-amine)}{CH3 - CH2 - CH2 - NH2}}}\]
APPEARS IN
संबंधित प्रश्न
Write the structure of the product formed when carboxylic acid is heated with a dehydrating agent like P2O5
Write the reducing agent which CANNOT reduce –COOH group.
What is the action of following on proponal?
Hydroxyl amine
What is the action of following on proponal?
Hydrazine
Write the preparation reactions for acid amide from the following.
Acid chloride
Write reactions for the following conversions.
4-Nitrobenzoic acid to Nitrobenzene
Write chemical reactions to convert –COOH group of acetic acid into the following.
C2H5OH
Write reaction for preparation of acetophenone from benzoyl chloride.
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be ____________.
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
Which of following elements does not form amide when reacted with ammonia?
In the following reaction:
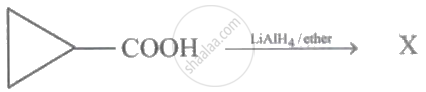
The product X is:
A flavouring agent found in oil of wintergreen is ______.
Products in the following reaction are:
\[\ce{CH3COOH + PCl3 ->[\Delta]}\]
Which of the following carboxylic acids will have the highest acidity?
The elimination of CO2 from a carboxylic acid is known as ____________.
What amount of dinitrogen contains 3.6 × 1018 molecules?
Which among the following is the strongest acid?
The oxidation number of B in NaBH4 is ____________.
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Which of the following carboxylic acids ha highest acidic strength?
Write structure of adipic acid.
Which of the following can reduce?
\[\ce{RCOOH -> RCH2OH}\]
How will you prepare Acetic anhydride from acetic acid?
