Advertisements
Advertisements
प्रश्न
Identify the gas evolved and give the chemical test in the following case:
Dilute hydrochloric acid reacts with iron (II) sulphide.
उत्तर
Gas Evolved:
When dilute hydrochloric acid reacts with iron (II) sulphide, the gas evolved is hydrogen sulphide.
Reaction:
\[\ce{FeS + 2HCl -> FeCl2 + H2S ^}\]
Chemical Test for Hydrogen Sulphide: To test for \[\ce{H2S}\] gas, you can use lead acetate paper:
- Moisten a strip of lead acetate paper.
- Expose it to the gas evolved.
Observation: If \[\ce{H2S}\] is present, the lead acetate paper will turn black due to the formation of lead sulphide \[\ce{(PbS)}\].
\[\ce{Pb(C2H3O2)2 + H2S -> PbS + 2CH3COOH}\]
APPEARS IN
संबंधित प्रश्न
State your observations when Concentrated Sulphuric acid is added to Sugar Crystals
Name the products formed when hot and concentrated sulphuric acid reacts with Copper.
Define the following term : Dehydrating agent
Give reason for the following:
When barium chloride is added to dilute sulphuric acid, a white precipitate is formed.
Give main difference between drying agent and dehydrating agent.
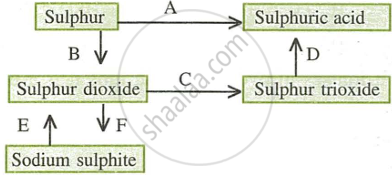
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Name the gas evolved in following case:
The gas produced by the action of concentrated sulphuric acid on sodium chloride.
Which property of sulphuric acid accounts for its use as a dehydrating agent ?
An acid obtained from concentrated nitric acid on reaction with Sulphur ______.
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
What is the property of sulphuric acid that makes it a suitable reagent for the reaction?
