Advertisements
Advertisements
प्रश्न
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
पर्याय
O2(g)
CH4(g)
CO2(g)
H2O(l)
MCQ
उत्तर
O2(g)
Explanation:
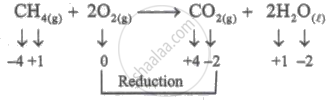
Since oxygen (O2) is undergoing reduction, it is oxidising agent.
shaalaa.com
Balancing Redox Reactions in Terms of Loss and Gain of Electrons
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
