Advertisements
Advertisements
Question
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
Options
O2(g)
CH4(g)
CO2(g)
H2O(l)
MCQ
Solution
O2(g)
Explanation:
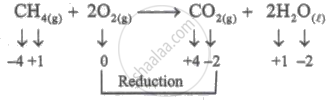
Since oxygen (O2) is undergoing reduction, it is oxidising agent.
shaalaa.com
Balancing Redox Reactions in Terms of Loss and Gain of Electrons
Is there an error in this question or solution?
