Advertisements
Advertisements
प्रश्न
Identify the terms for the following:
The experiment which demonstrates high solubility of ammonia gas.
उत्तर
Fountain Experiment
Explanation:
The high solubility of ammonia gas in water can be demonstrated by performing the fountain test in which a fountain of litmus solution is obtained when ammonia dissolves in water.
APPEARS IN
संबंधित प्रश्न
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
How will you demonstrate the solubility of ammonia in water? Explain.
The diagram shows a simple arrangement of the fountain experiment:
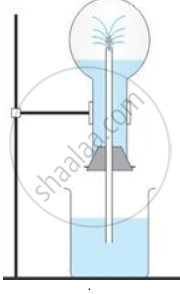
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.

- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonia in excess is mixed with chlorine.
Differentiate between:
Aqueous ferrous and ferric sulphate solution
Write an equation to illustrate the reducing nature of ammonia.
Of the two gases, ammonia and hydrogen chloride, which is denser? Name the method of collection of this gas.
