Advertisements
Advertisements
प्रश्न
If the relative molecular mass of A is 90, what is the molecular formula of A?
उत्तर
The relative molecular mass of A is 90
Empirical Formula mass of compound is = 45
Then, n = Molecular mass/ Empirical Formula Mass
= 90/45 = 2
Molecular formula of compound = n x Empirical formula
= 2 x (CO2H) = C2O4H2.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
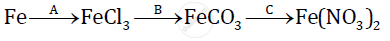
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of potassium nitrite formed.
(K = 39, 0 = 16, N = 14)
An organic compound has the following percentage composition: C = 12.76%, H = 2.13%, Br = 85.11%. The vapour density of the compound is 94. Find out its molecular formula.
Find the total percentage of oxygen in magnesium nitrate crystal Mg(NO3)2.6H2O.
[H = 1, N = 14, O = 16, Mg = 24]
Calculate the percentage of phosphorous in the fertilizer superphosphate, Ca(H2PO4)2. [Ca = 40, H =1, P =31, O = 16] (Correct to 1 decimal place)
Calculate the percentage of platinum in ammonium chloroplatinate (NH4)2PtCl6.
[N = 14, H = 1, Pt = 195, Cl =35.5]
(Give your answer correct to the nearest whole number)
Calculate the percentage of sodium in sodium aluminium fluoride (Na3AIF6).
[F = 19, Na = 23, Al = 27]
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Calculate the relative molecular mass of:
CH3COONa
The following question refers to one mole of chlorine gas.
If the relative atomic mass of chlorine is 35.5, what will be the mass of 1 mole of chlorine gas?
