Advertisements
Advertisements
प्रश्न
If the internal energy of an ideal gas U and volume V are doubled then the pressure ____________.
पर्याय
doubles
remains same
halves
quadruples
उत्तर
If the internal energy of an ideal gas U and volume V are doubled then the pressure remains same.
APPEARS IN
संबंधित प्रश्न
Find the kinetic energy of 3 litre of gas at S.T.P given standard pressure is 1.013 × 105 N/m2.
Determine the pressure of nitrogen at 0°C if the density of nitrogen at N.T.P. is 1.25 kg/m3 and R.M.S. speed of the molecules at N.T.P. is 489 m/s.
The ratio γ = `"C"_"p"/"C"_"v"` for a gas mixture consisting of 8 g of helium and 16 g of oxygen is ____________.
Which of the following shows the correct relationship between the pressure and density of an ideal gas at constant temperature?
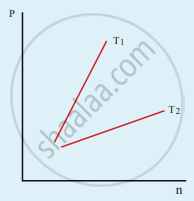
The following graph represents the pressure versus number density for an ideal gas at two different temperatures T1 and T2. The graph implies
Estimate the total number of air molecules in a room of a capacity of 25 m3 at a temperature of 27°C.
According to the assumptions made in the kinetic theory of gases, when two molecules of a gas collide with each other, then ______.
Derive an expression for the pressure exerted by a gas on the basis of the kinetic theory of gases.
Does an ideal gas exist in practice?
The velocities of five molecules are 2 m/s, 3 m/s, 4 m/s, 5 m/s and 6 m/s. Find the root mean square velocity of molecules.
