Advertisements
Advertisements
Question
If the internal energy of an ideal gas U and volume V are doubled then the pressure ____________.
Options
doubles
remains same
halves
quadruples
Solution
If the internal energy of an ideal gas U and volume V are doubled then the pressure remains same.
APPEARS IN
RELATED QUESTIONS
A sample of an ideal gas is at equilibrium. Which of the following quantity is zero?
The ratio γ = `"C"_"p"/"C"_"v"` for a gas mixture consisting of 8 g of helium and 16 g of oxygen is ____________.
Which of the following shows the correct relationship between the pressure and density of an ideal gas at constant temperature?
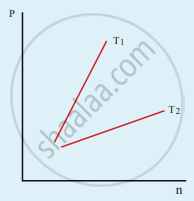
The following graph represents the pressure versus number density for an ideal gas at two different temperatures T1 and T2. The graph implies
What is the microscopic origin of pressure?
A perfect gas of 'N' molecules, each of mass 'm', moving with velocities 'C1', 'C2', ...... .'CN' is enclosed in a cubical vessel of volume 'V'. The pressure exerted by the gas on the walls of the vessel is ______. ('p' = density of gas)
According to the assumptions made in the kinetic theory of gases, when two molecules of a gas collide with each other, then ______.
Derive an expression for the pressure exerted by a gas on the basis of the kinetic theory of gases.
What is an ideal gas?
Temperature remaining constant, if you double the number of molecules in a box, the pressure will ______.
