Advertisements
Advertisements
प्रश्न
In the extraction of aluminium: Draw the diagram for the extraction of aluminium.
उत्तर
Extraction of aluminium:

APPEARS IN
संबंधित प्रश्न
Name two metals which are found in nature in the free state.
In the extraction of aluminium: Why is it necessary to replace anodes time to time?
Name one ore of copper (other than cuprite). Which compound of copper is present in this ore? Also, write its chemical formula.
Name the metal which is extracted from the ore called 'rock salt'.
An ore of manganese metal is:
(a) bauxite
(b) haematite
(c) cuprite
(d) pyrolusite
Give the principles of the hydrolytic method.
Distinguish between ‘roasting’ and ‘calcination’. Which of these two is used for sulphide ores and why?
Name the following:
Two metals which are found in the free or native state.
Name the following:
The mixture of materials fed into a furnace to extract a metal.
Complete the incomplete statement with missing words:
Metals are ______ while non-metals are ______ conductors of heat.
How many valence electrons are present in non- metals?
Name the methods by which concentrated ore is converted to metallic oxide.
Observe the given figure of reactivity series of metals and answer the following questions:

Reactivity series of metals
- Name two metals which react with water.
- Name two moderately reactive metals.
- Name the most highly reactive metal and the most less reactive metal.
If Cu, Fe, Zn, Al elements are arranged in increasing order of their reactivity then the correct order would be which of the following?
Explain concept with example/explain with the help of a balanced equation.
Calcination
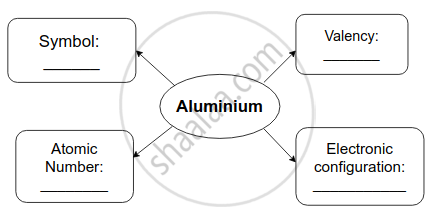
Complete flow chart given below.
Read the following passage and answer the questions.
According to the reactivity series, zinc is more reactive than iron, iron is more reactive than silver. During study of this, a student dipped the iron nails in silver nitrate solution.
- What is reactivity series?
- What will happen when iron nails are dipped in silver nitrate solution?
- Which type of reaction happens when iron metal reacts with silver nitrate solution?
- What will happen if a zinc rod is used instead of iron nail?
The highly reactive metals like Sodium, Potassium, Magnesium, etc. are extracted by the ____________.
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
