Advertisements
Advertisements
प्रश्न
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
उत्तर
Phenoxide ion is more reactive than phenol towards electrophilic substitution. The negative charge on oxygen is transferred to benzene through resonance. This helps in the attachment of CO2 which is a weak electrophile to the benzene ring finally giving salicylic acid.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction:
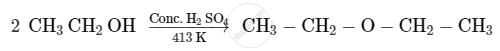
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
The compound which reacts fastest with Lucas reagent at room temperature is:
Dehydration of 2-butanol yields:
Which of the following is not true in case of reaction with heated copper at 300°C?
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
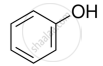
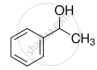
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Write the mechanism of acid dehydration of ethanol to yield ethene.
