Advertisements
Advertisements
Question
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Solution
Phenoxide ion is more reactive than phenol towards electrophilic substitution. The negative charge on oxygen is transferred to benzene through resonance. This helps in the attachment of CO2 which is a weak electrophile to the benzene ring finally giving salicylic acid.
APPEARS IN
RELATED QUESTIONS
Write the mechanism of the following reaction:
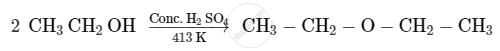
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Lucas reagent is ____________.
By which of the following methods alcohol can be prepared in excellent yield?
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 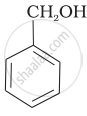 |
| (b) | 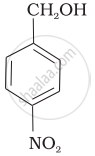 |
| (c) | 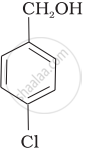 |
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
