Advertisements
Advertisements
Question
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Solution
The C – O – H bond angle in alcohols is slightly less than the tetrahedral angle (109.5°) because of larger repulsions between the lone pairs of electrons. For example in methanol C – O – H bond angle is 108.9°.
In ethers, the C – O – C bond angle is slightly greater than tetrahedral angle. For example in dimethyl ether C – O – C bond angle is 111.7°. The larger bond angle in ethers may be because of greater repulsions between bulkier alkyl groups as compared to one H in alcohols.
APPEARS IN
RELATED QUESTIONS
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Lucas test is used for the detection of _____________.
Which of the following is not true in case of reaction with heated copper at 300°C?
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 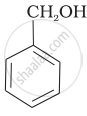 |
| (b) | 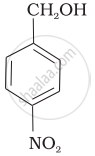 |
| (c) | 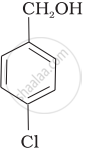 |
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
